The door to CLINUVEL’s expansion towards a diversified and sustainable healthcare company was opened by the approval of SCENESSE® (afamelanotide 16mg) in October 2019 by the US Food and Drug Administration (FDA) for adult erythropoietic protoporphyria (EPP) patients. After decades of misfortune, CLINUVEL’s reviewed strategy to develop a novel melanocortin for an unmet medical need had then been validated by the two leading regulatory bodies of the world in 2014 and 2019, enabling the Group to evaluate how it could expand its operations and address broader audiences.
Strategy Reset
The initial unsuccessful strategy of the Company to the end of 2005 was to develop a drug to assist tanning of the skin as a lifestyle product. Whilst a large recreational market was receptive to such a drug, its more cosmetic than medicinal purpose was never going to be supported by the regulators and CLINUVEL thus had to come up with a brand new strategy.
In November 2005, Dr Philippe Wolgen was appointed CEO of CLINUVEL. Together with the support of the Board and a revitalised management team, the Company’s strategy was reset to focus on the development and commercialisation of one drug for one indication with a high unmet medical need. This strategy met numerous obstacles and was implemented over 15 years by one homogenous team. A number of indications were researched before the genetic metabolic disorder EPP was selected as the lead indication with which to prove the novel concept of systemic photoprotection. CLINUVEL completed clinical studies over several years and built expertise in melanocortins and their role in the human body, as well as the impact of light on human biology.
"The door not only opened for SCENESSE® to treat EPP patients in the USA in October 2019, but also unlocked the opportunity to expand the research and development program to assess the application of SCENESSE® to other indications."
The Company had commenced first distribution of SCENESSE® for EPP patients in Italy in 2010 and Switzerland in 2012 under special access programs, while in 2014 the European Medicines Agency (EMA) approved SCENESSE® for adult EPP patients. Commercial operations began in the European Union in June 2016 after agreement was reached on a rigorous risk management plan to monitor and report the patient experience with SCENESSE®. Liaison continued with the FDA on regulatory approval in the USA, with the FDA seeking real world evidence from the European programs. In October 2019, the FDA granted approval to CLINUVEL to distribute SCENESSE® in the USA and commercial operations commenced in April 2020.
Success of the strategic reset of 2005, is reflected by:
- the increasing number of EPP patients being treated who report a positive safety experience over time; and
- the financial performance of the Company, achieving record revenues and profit in June 2021, in the fifth consecutive year of positive cashflow and profit.
The Company has been and remains focused on the safety of the melanocortin first-in-class drug. The positive real-world patient experience builds confidence as to the implemented strategy and together with the expertise built in melanocortins, enables the expansion of research and development activities to treat a much wider range of indications and broader audiences.
Regulatory Approvals Unlock CLINUVEL’s Expansion
The key regulatory approvals that have been granted reflect the acceptable safety profile of SCENESSE®, the world’s first systemic photoprotective drug. This position was reached after many years of hard work to build credibility as a pharmaceutical company with expertise in melanocortins. Subsequent to the EMA and FDA approvals, the Australian Therapeutic Goods Administration and Israeli Ministry of Health granted approval to SCENESSE® for adult EPP patients in October 2020 and February 2021, respectively.
The door was not only unbolted for SCENESSE® to treat EPP patients in the USA in October 2019, but also unlocked the opportunity to expand the research and development program to assess the application of SCENESSE® to other indications.
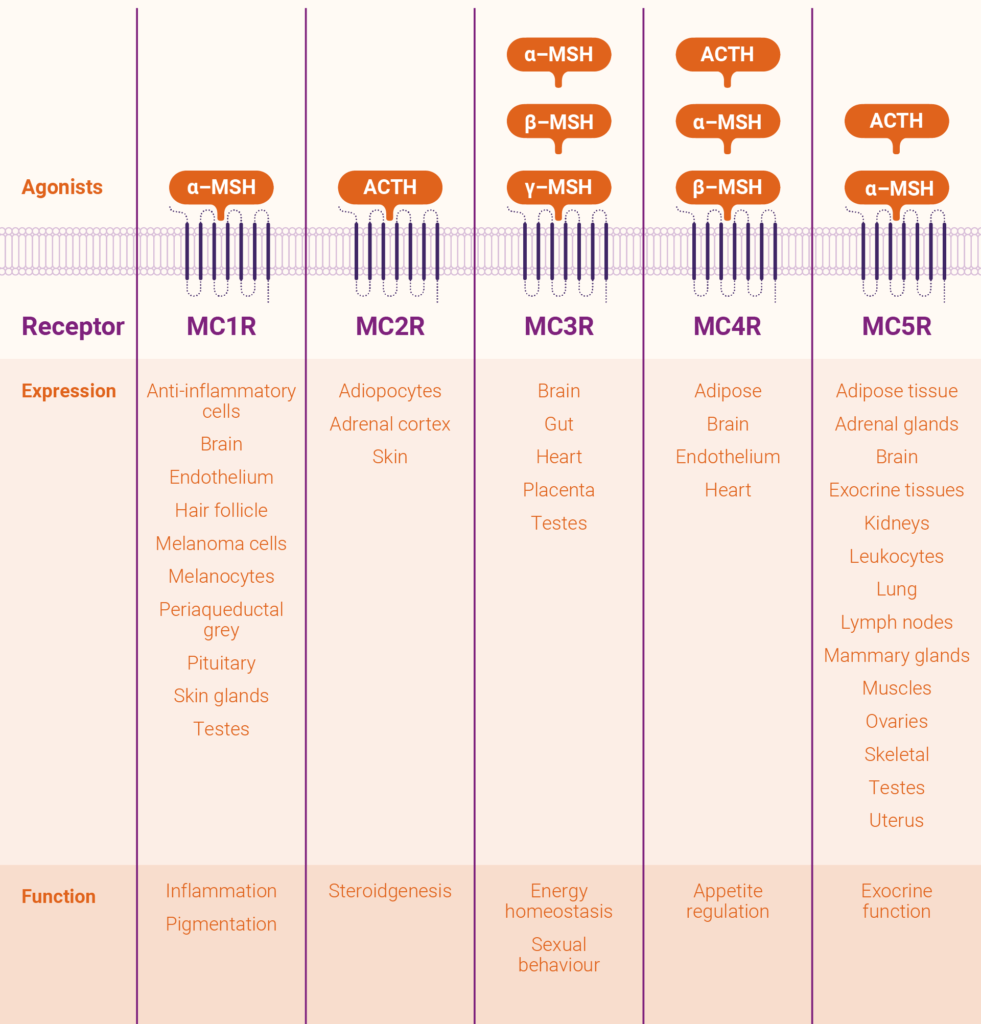
We know from our own work, and an increasing dossier of worldwide research, that melanocortins can play a multifactorial role in the body, binding with one or more of the five melanocortin receptors (MC1R to MC5R) to influence a range of physiological activities. These range from photoprotection, repigmentation and DNA repair of the skin, to inflammation, energy homoestasis, appetite and sexual function. The table shows a global snapshot of the distribution of the five melanocortin receptors and the functions they influence.
From a foundation built on expertise and experience, CLINUVEL has progressed to allocate resources to target other unmet medical needs. The intention is to assess the ability of afamelanotide to bind with different melanocortin receptors and assist different functions of the human body. Afamelanotide is a synthetic hormone of the naturally occurring alpha-melanocyte stimulating hormone (α-MSH), which binds to receptors showing affinities to MC1R, MC3R, MC4R and MC5R. In addition to progressing our commitment to develop a repigmentation treatment for vitiligo, we have expanded the research and development program to DNA Repair – with an initial focus on xeroderma pigmentosum (XP) – and to stroke. These indications provide the opportunity to treat more patients and build upon our commercial success. The Company’s focus on the research and development, regulatory approval, and commercialisation of treatments for these indications is justified based on the acceptance and status of SCENESSE® as a safe treatment for EPP.
The expertise developed in melanocortins will also be extended to non-pharmaceutical products for broader audiences within the general population at risk from ultraviolet (UV) and high energy visible (HEV) light. We assess that dermatocosmetic products are needed by these underserved groups.
The next article covers how we are organising the Company to meet the challenges of the future. In subsequent articles of the Annual Report, we round out the activities and initiatives to build a diversified and sustainable group of companies with:
- an update on the growth of commercial operations based on SCENESSE® for EPP;
- details of the expansion of the research and development program and translation of our technology to targeted indications; and
- outline the rationale and plans for a dermatocosmetic product range to assist individuals at high risk of exposure to light.