Long-term Value of Integration and Diversification
CLINUVEL’s business model strives to build and retain key pharmaceutical functions in-house, rather than outsource to external business partners and suppliers. The overall aim is to further integrate functions while diversifying revenue streams. The rationale of this approach is found in the Company’s objectives for independence with self-determination and control of quality, costs and supply, reflecting the Company’s resourceful culture.CLINUVEL undertakes several functions in-house:
- Formulation R&D, including select pre-clinical development;
- Design and conduct of clinical studies;
- Regulatory affairs, pharmacovigilance and quality assurance activities, including key manufacturing activities;
- Direct distribution of SCENESSE® to European EPP Expert Centres and US Specialty Centers;
- Pricing and reimbursement functions, including patient support and the SCENESSE® Assistance Program in the USA;
- Finance, facilitated by preservation of cash balances to allow for planned organic and inorganic growth and expansion; and
- Communications, branding and marketing, and investor relations.
Plans to diversify the Company are being executed and aim to result in new revenue streams as treatments are provided for indications with larger patient populations than EPP. In other words, the firm’s efforts go towards spreading the Company’s activities, products and markets. This strategy is one to diversity revenue streams, part of a risk management plan. The risk of the chosen strategy lies in its implementation, which is mitigated by a deliberate and thorough approach of the Company to expand its activities based on its existing expertise in melanocortins and photomedicine, and central nervous system. The Group has increased its workforce to support its activities accordingly.
Infrastructure and Know-How to Support Expansion
CLINUVEL has built enhanced infrastructure and know-how in support of its expansion plans.
With this overview of CLINUVEL’s strategic approach in mind, more specific plans in EPP patient treatment, drug development and indications, and PhotoCosmetics, are outlined below.
Enabling EPP Patient Treatment
CLINUVEL continues to focus on the distribution of SCENESSE® for EPP patients in existing jurisdictions to meet patient and physician demand. In this regard, work with external contract manufacturers and raw material suppliers continues to ensure product supply at current Good Manufacturing Practice standard.
Europe
As part of the conditions attached to European marketing authorisation, CLINUVEL will maintain its commitment to the European EPP Disease Registry to monitor the long-term safety of SCENESSE®.
The Company recognises the impact of EPP on patients under the age of 18 and the potential for SCENESSE® as a treatment for adolescent patients. CLINUVEL subsequently proposed an expansion of the drug’s label to the European Medicines Agency (EMA) to include adolescent patients and expect to provide an update later in 2023.
USA
CLINUVEL aims to establish a network of 120 Specialty Centers across the US to provide greater treatment access for EPP patients. This network may also play a role in the clinical development of SCENESSE® for other indications. Work continues to provide treatment access for all US EPP patients.
Other Jurisdictions
The Company is committed to gaining market access for SCENESSE® in other jurisdictions around the world.
Melanocortin Drug Development
CLINUVEL is a world leader in melanocortins, evaluating a number of melanocortin-based pharmaceuticals for a range of patient groups with unmet need.
CLINUVEL expects progress across several clinical programs with afamelanotide:
- DNA Repair Program – results of the first two XP studies (CUV156 and CUV152) and the study on healthy volunteers (CUV151) are expected in FY2024. Subject to these results, additional studies will be undertaken with the objective of completing a dossier of patient results of sufficient gravitas to present to the EMA to extend the approved label of SCENESSE® to XP patients.
- Vitiligo – the results of CUV104 will be available in FY2024 and CUV105, a new, larger study, using SCENESSE® in combination with NB-UVB phototherapy, will commence as soon as design and ethics approvals are received. The objective is to submit a dossier to the US FDA to consider approval to extend the label of SCENESSE® to vitiligo patients.
- Variegate porphyria – first efficacy results of CUV040 are expected in 2024. After assessing the results, the next steps will be determined, including the need for another study, with a view to substantiate a submission to the EMA to extend the label of SCENESSE® to VP patients in Europe.
- Stroke – first efficacy results of CUV803 are expected in FY2024. The path to regulatory approval of afamelanotide for Stroke is a longer term one. Additional and larger studies will need to be undertaken before regulatory approvals can be sought and a timeframe to commercialisation of at least 5 years is considered likely.
Clinical studies in other indications are planned to be announced in 2024 and 2025.
PhotoCosmetic Products
CLINUVEL is establishing a new category of products as PhotoCosmetics, with the objective of making the brand a household name by 2026.
CYACÊLLE is planned for wider launch in new jurisdictions, particularly the USA. The PhotoCosmetic products for DNA repair and melanogenesis will continue to be developed by the Research, Development & Innovation Centre in Singapore, for planned launch in 2024 and 2025, respectively.
Regular Bulletins from the CBM team, addressing different aspects of CLINUVEL’s brand architecture, will be issued to provide updates to stakeholders.
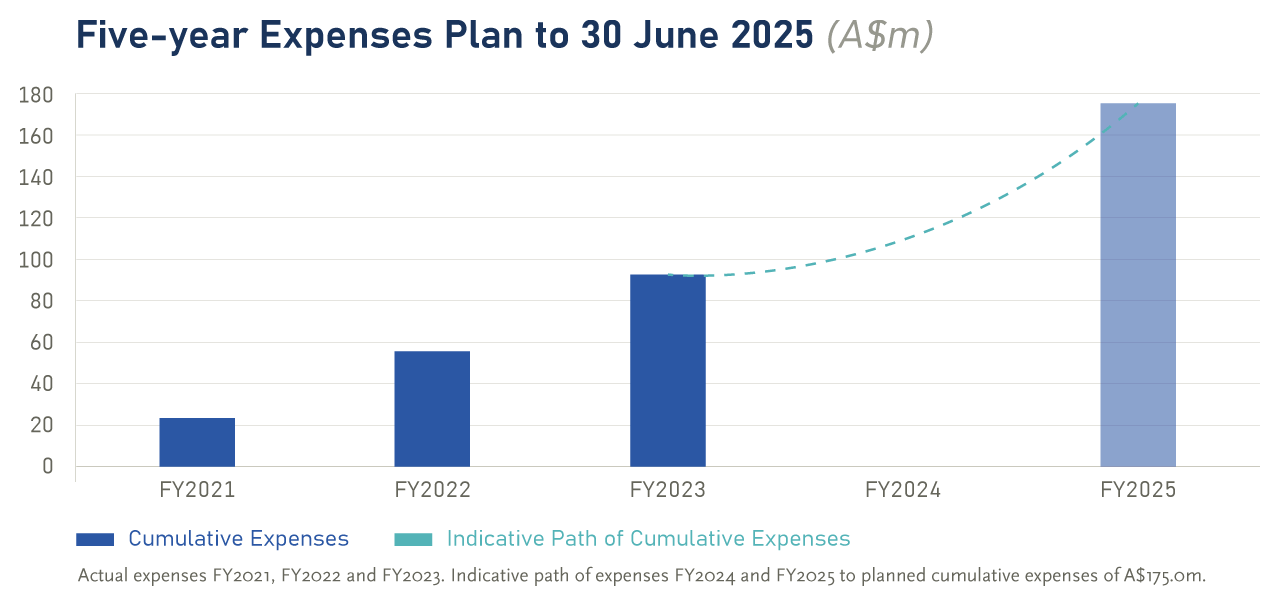
Cumulative Expenses Plan
We have provided a five-year projection of expenses we expect to incur to aim for our objectives. Excluding capital expenditures and marketing expenses on the PhotoCosmetics initiative, expenses of $175.0 million are planned over the five years to the end of FY2025.To date, expenses incurred in FY2021 to FY2023 total $92.8 million, or 53% of the expenses plan. The chart below shows the indicative path to achieve this projection.
Acquisitions
CLINUVEL has long expressed an interest in acquiring assets to augment the planned organic growth of the Company. An acquisition needs to fit several criteria:
- An existing management team with complementary skills and willingness to integrate within the Group;
- Scope to add value from its experience and expertise;
- Complement or add to our activities;
- In either a late clinical stage biotech; and/or a
- Manufacturer that advances our objectives.
CLINUVEL does not rule out recourse to external finance – whether this be bank debt and/or a capital raising – to accelerate or complete one or more acquisitions. The option to secure finance to allow for an acquisition is one of several avenues to grow the Group inorganically. The ongoing monitoring and assessment of opportunities does not engage more management time and effort than warranted by the potential value add of the appropriate incorporation.
Primary Listing of the Company
The primary listing of CLINUVEL is the Australian Securities Exchange (ASX) – since 2001. We have made it explicit in our recent communications that consideration would be given to a primary listing elsewhere if the benefits to the Group and stakeholders are compelling. We have also stated that we are not inclined to a dual listing due to the duplication of compliance and reporting costs, and dilution of trading volume. Therefore, our approach is to either remain on the ASX or move to a listing in another country, if opportunities are sufficiently compelling. The implications of a change in listing will be thoroughly assessed such that shareholders benefit and do not lose out or suffer negative consequences.
Summary
CLINUVEL is on the path towards a highly integrated and diversified pharmaceutical company. Its transformation will be reflected in four pharmaceutical products for eight indications – four in photomedicine and four in CNS conditions – and three PhotoCosmetic product lines which will make CLINUVEL a household name. CLINUVEL will be recognised as an expert in melanocortins and their therapeutic role in managing conditions of the skin and brain. The benefit of this integration and diversity will be a sustainable business able to perform with resilience and dynamism over the long-term.