Plans 2023 and Beyond
Objective and Focus
CLINUVEL is building a diversified and sustainable pharmaceutical company based on the specialisation in melanocortins. Both the Board and management believe this will generate significant incremental long-term value.
Integrated Business Model
A key factor in CLINUVEL’s success to date has been the integrated business model employed to manage the diversity of its activities. This means the conduct of key functions of the business are undertaken ‘in-house’ where practical. These functions are typically outsourced by other pharmaceutical companies.
This approach has been beneficial to shareholders. CLINUVEL’s direct distribution of SCENESSE® to EPP Expert Centres in Europe and Specialty Centers in the USA means margins are kept intact by our self-distribution model. Other activities managed ‘in-house’ – such as formulation R&D, regulatory affairs, and quality assurance – also result in better control of outcomes and costs. The integrated business model is also reflected in the establishment of the Communications, Branding & Marketing Division to reach the targeted audiences at highest risk of exposure to UV and HEV light.
An integrated business model also fits with CLINUVEL’s culture of self-determination, resourcefulness, and persistence, and serves to provide an internal environment in which supports the development of well rounded, highly skilled, and capable professionals.
Following the easing of restrictions imposed at the height of the COVID-19 pandemic, we can reflect on supply arrangements and determine that independence of operation has many advantages compared to a broader model based on outsourcing and consequent dependence on suppliers. This is also the rationale for the establishment of the Manufacturing Division in which CLINUVEL will manufacture its own products and, in time, offer its manufacturing capabilities to other pharmaceutical enterprises. The divisional structure of CLINUVEL has evolved to support the integrated business model and as shown below; this encompasses the Pharmaceutical and Healthcare Solutions Divisions, as well as divisions focussed on Communications, Branding and Marketing and Manufacturing. The research and development activities of the Group are undertaken by the VALLAURIX Research, Development and Innovation Centre in Singapore.
We will continue to develop this integrated business model to support the ongoing and future activities of the Group.
Plans
Our plans for FY2023 and beyond in each functional area of the business are shown below:
Increase EPP patient access to SCENESSE® treatment
In existing jurisdictions (Europe, USA, Israel) and new jurisdictions (refer text box) and extend the use of afamelanotide to adolescent EPP patients.
Expand the product range
This encompasses the expanded pharmaceutical drug portfolio, as well as the translation of our melanocortin expertise and know-how to dermatocosmetic products, as detailed in the preceding feature of the Annual Report.
Continue to progress the clinical program
We will continue the existing studies already outlined through 2022 and into 2023. We also expect to announce new indications that will result in further expansion of the clinical program.
Progress the Manufacturing Division
Plans to establish self-manufacturing are at an early stage and developments will be shared with stakeholders as they progress.
In addition to the above, the Group will consider opportunities for inorganic growth that add value to the Group’s capabilities and achievement of its objectives.
New jurisdictions planned for distribution of SCENESSE® for EPP
- Australia – we are progressing an application to have SCENESSE® listed on the Pharmaceutical Benefits Scheme for reimbursement of the cost of treatment of Australian EPP adult patients.
- China – we are collaborating with a local partner to treat EPP patients as part of a named patient program. This will provide Chinese patient data to combine with European and US patient data to submit to Chinese authorities for consideration of approval to distribute SCENESSE® to EPP patients in China.
- Japan – we are assessing the dossier that the authorities require to assess approval of the distribution of SCENESSE® to EPP patients in Japan.
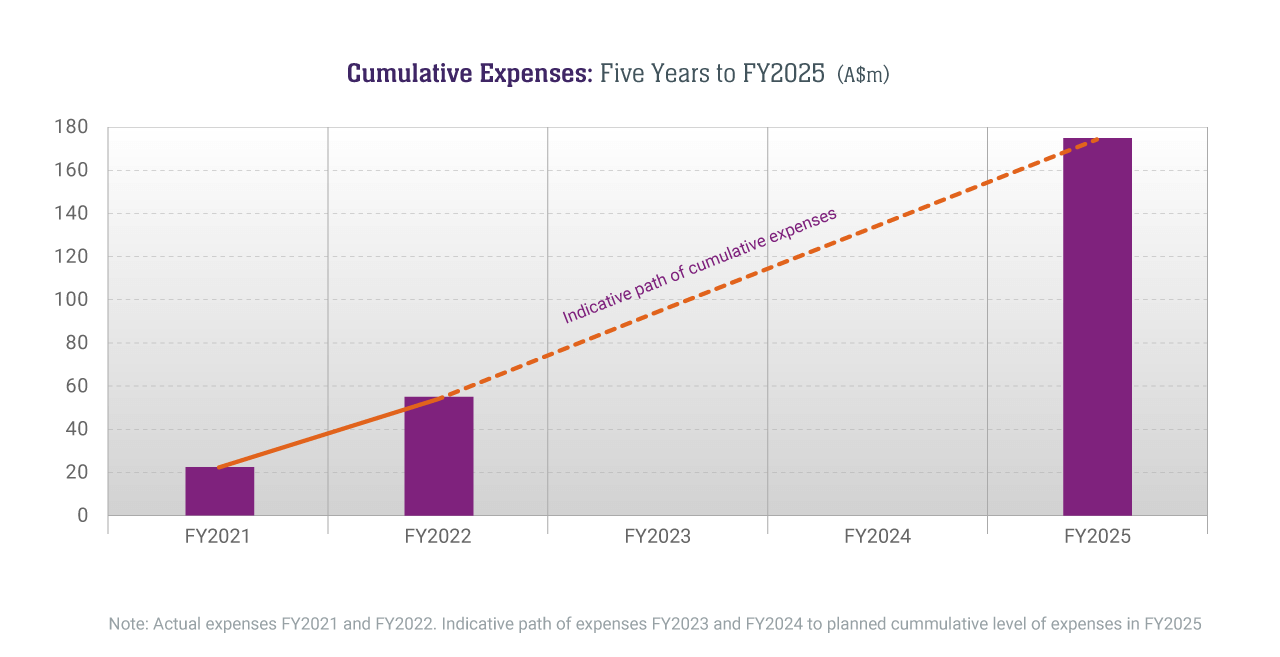
Expenditures to Rise to Support Growth
We have provided a five-year guide of the expenses expected to achieve our plans. Excluding capital expenditures, expenses of A$175 million are planned over the five years to the end of FY2025. To date, the expenses incurred in FY2021 (A$22.738 million) and FY2022 (A$32.667 million) put us on track to achieve this projection.
Summary
These plans detail a clear and deliberate growth strategy to add significant long-term value to the Group. We are persistent and determined and believe in the ability of the melanocortin family of hormones to have influence on the quality of the lives of many people, whether this be through pharmaceutical drug treatments or dermatocosmetic products. This is the course we at CLINUVEL have embarked upon and we welcome all existing and potential stakeholders to this journey.