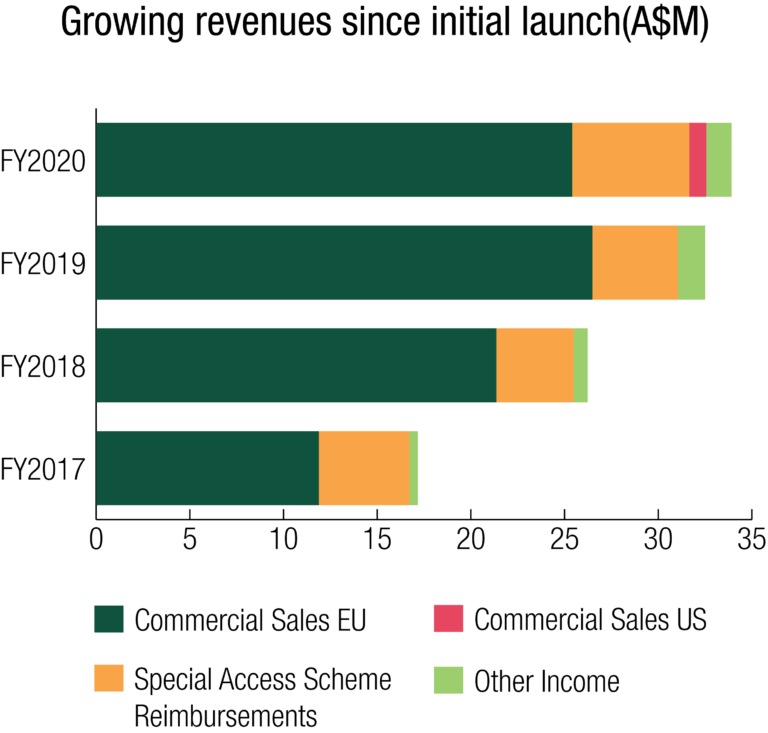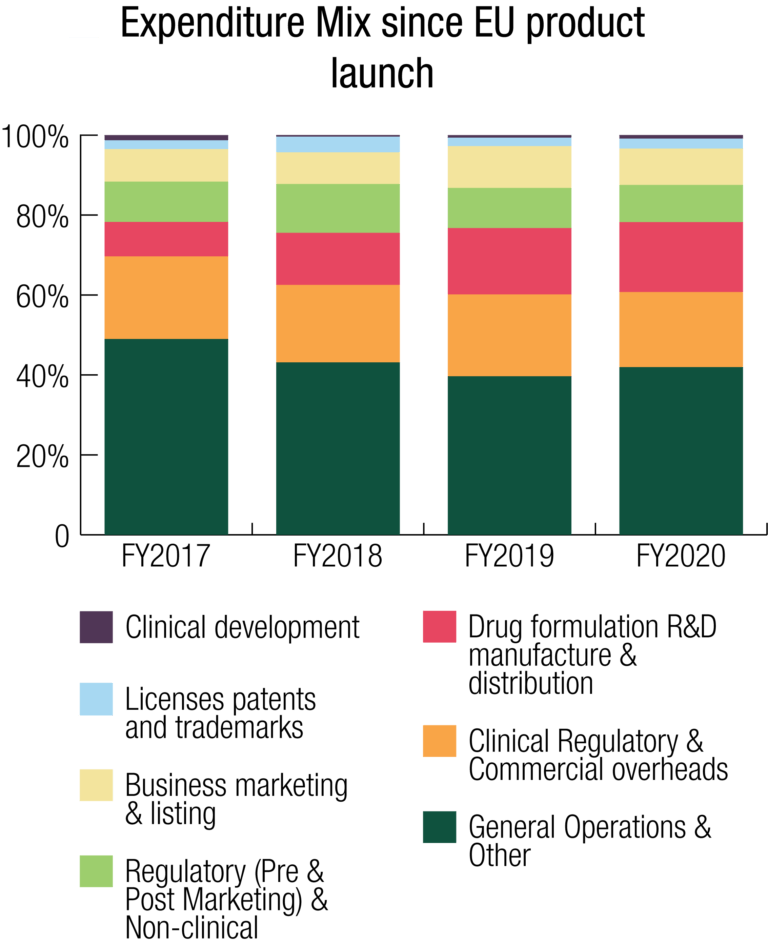SINGAPORE RESEARCH, DEVELOPMENT & INNOVATION CENTRE
DIRECTORS’ REPORT
DIRECTORS’ REPORT
The Directors of the Board present their report on the Company for the financial year ended 30 June 2020 and the Auditor’s Independence Declaration thereon.
DIRECTORS
The names of Directors in office during or since the end of the year are set out below.

WILLEM BLIJDORP
Non-Executive Director, Funda
Appointed 21 January 2015, Chair since 30 November 2019
Background
Mr Blijdorp is an internationally recongnised entrepreneur who has helped build the B&S Group, one of the largest global trading houses, in a period spanning three decades. Mr Blijdorp has led B&S’s growth, with the Dutch group focused on specialty distribution services to difficult to serve markets. The B&S Group has global reach and is a leader in its market sector.
Formerly B&S Group’s CEO, Mr Blijdorp now serves on its Supervisory Board and is a majority shareholder, focussing on the Group’s development and expansion strategy. He led and oversaw the Group’s initial public offering on Euronext Amsterdam in March 2018.
In 2014 Mr Blijdorp was recognised for his expertise in merger and acquisitions and commercial leadership as the Ernst & YoungbEntrepreneur of the Year in the Netherlands, and runner-up in its European Union awards. Since becoming a director of CLINUVEL in 2015, Mr Blijdorp has provided a valuable contribution to setting the Group’s long-term strategy for product commercialisation, growth, and future plans to further diversify CLINUVEL.
Relevant Skills
- entrepreneurship, commercial prowess
- general management
- financial management
- experienced in listed company Directorships
Committee Membership
Chair of the Remuneration Committee
Chair of the Nomination Committee
Member of the Audit and Risk Committee
Current Directorships and other interests
Director of the Supervisory Board of the B&S Group (the Netherlands)
Relevant interest in Shares and performance rights
Shares: 1,743,118
Performance Rights: –
Other listed company Directorships (last 3 years)
None
PHILIPPE WOLGEN
Chief Executive Officer, MBA, MD
Appointed to Board 1 October 2005, appointed Chief Executive Officer 28 November 2005
Background
Under Dr Wolgen’s leadership since late 2005, a long-term strategy for CLINUVEL was devised. The lead product SCENESSE® (afamelanotide 16mg) was reformulated, its medical application identified, European marketing authorisation was obtained in 2014 and distributed in the European Economic Area from June 2016. Dr Wolgen oversaw the submission of the scientific dossier to the US Food & Drug Administration (FDA) under a New Drug Application, which was approved in October 2019. First treatment of US patients commenced in April 2020. SCENESSE® is the first melanocortin drug to have completed a clinical trial program and obtain marketing authorisation in two major markets.

Dr Wolgen has been instrumental in the Company’s corporate turnaround, rebuilding a share register of long-term professional and institutional investors. He led CLINUVEL to attract more than AU$110 million in investments, his international contacts and network contribute to the strategic support CLINUVEL enjoys globally.
Under his tenure a business model was adopted to develop and launch SCENESSE®, guiding the Group through a complex pharmaceutical product development program. His overall business execution and exact financial management is viewed as exemplary within the life sciences industry and the funding strategy he led is considered unique within the sector.
Dr Wolgen is currently leading the Group’s expansion, with an immediate focus on the US and the further development of the product pipeline for various market segments. His focus has been to establish a professional management team to execute the corporate objectives set and prepare the next generation of managers.
Dr Wolgen’s long track record speaks to a strongly focussed, competitive and conscientious professional who is known to persevere in meeting challenging business objectives. He holds an MBA from Columbia University, NY. Trained as a craniofacial surgeon, Dr Wolgen obtained his MD from the University of Utrecht, the Netherlands.
Relevant Skills
- pharmaceutical research & development, commercialisation
- clinical expertise
- commercial knowhow, entrepreneurial outlook
- executive management, corporate turnarounds
- financial management
- capital market understanding
- experienced in listed company Directorships
Current Directorships and other interests
None
Other listed company Directorships (last 3 years)
None
Relevant interest in Shares and performance rights
Shares: 3,504,696
Performance Rights: 1,513,750*
*Performance Rights were issued to Dr Wolgen on 26 August 2020, consequent to shareholder approval at the 2019 AGM

BRENDA SHANAHAN
Non-Executive Director, BComm, FAICD, ASIA
Appointed 6 February 2007
Background
Mrs Shanahan is a pioneer in the Australian finance community. The first female stockbroker, Mrs Shanahan has also spent more than two decades working and investing in medical R&D and commercialisation. She is currently a non-executive director of Phoslock Environmental Technologies Ltd (ASX: PET). Mrs Shanahan is also a non-executive director of DMP Asset Management Ltd and SG Hiscock Ltd, a director of the Kimberly Foundation of Australia Ltd, and Chair of the Aikenhead Centre for Medical Discovery in Melbourne.
Previously Mrs Shanahan was a member of the Australian Stock Exchange and an executive director of a stockbroking firm, a fund management company and an actuarial company. Until 2017, she was Chair of St Vincent’s Medical Research Institute and also a non-executive director of Challenger Limited (ASX: CGF). Mrs Shanahan was formerly Chair of Challenger Listed Investments Ltd, the reporting entity for four ASX listed firms and formerly a non-executive director of Bell Financial Group (ASX: BFG). Mrs Shanahan also has served on and chaired various Audit and Risk Committees throughout her career, including Challenger Financial Services Group Ltd, Bell Financial Group, Victoria University, JM Financial Group Ltd, SA Water, AWB International Ltd, BT Financial Group and V/Line Passenger. She is the current Chair of of the Audit Committee for Phoslock Environmental Technologies Ltd (ASX: PET).
Mrs Shanahan joined CLINUVEL in 2007, and was Non-Executive Chair of the Board from late 2007 until July 2010. Her depth of experience across global markets and medical research provides significant value to the current Board and Group.
Relevant Skills
- research & development in life sciences
- capital market understanding
- executive management
- experienced in listed company Directorships
Committee Membership
Chair of the Audit and Risk Committee
Member of the Nomination Committee
Current Directorships and other interests
Chair of the Aikenhead Centre for Medical Discovery, Melbourne
Director of SG Hiscock Ltd
Director of DMP Asset Management Ltd
Director of Kimberly Foundation of Australia Ltd
Other listed company Directorships (last 3 years)
Phoslock Environmental Technologies Ltd (ASX: PET, since 2017)
Bell Financial Group (ASX: BFG, from 2012 to 2018)
Challenger Limited (ASX: CGF, from 2014 to 2017)
Relevant interest in Shares and performance rights
Shares: 258,969
Performance Rights: 25,000
KAREN AGERSBORG
Non-Executive Director, MD
Appointed 29 January 2018
Background
Dr Agersborg is a Board-Certified Endocrinologist in Pennsylvania, USA, currently serving as Clinical Endocrinologist at Easton Hospital, Steward Health, specialising in Endocrinology, Diabetes & Metabolism. Dr Agersborg had previously worked at Reading Hospital, West Reading and at Suburban Hospital, Norristown as Clinical Endocrinologist and served as Chief, Endocrinology, Diabetes, Metabolism at Chestnut Hill Hospital.

Dr Agersborg had an extensive career in managing commercial sales & distribution at Wyeth Pharmaceuticals (formerly Ayerst Laboratories). Dr Agersborg has played an integral role in setting the CLINUVEL Group’s US commercial strategy, resulting in the US FDA’s approval of SCENESSE® in October 2019.
Relevant Skills
- pharmaceutical research & development, commercialisation
- relevant knowledge on melanocortins, clinical expertise
- commercial knowhow in US pharmaceuticals
- general management
- experience in private company Directorships
Committee Membership
Member of the Remuneration Committee
Member of the Nomination Committee
Current Directorships and other interests
Member of the American Osteopathic Association
Fellow of the American Association of Clinical Endocrinologists
Fellow of the American College of Osteopathic Internists.
Doctorate of Osteopathic Medicine
Other listed company Directorships (last 3 years)
None
Relevant interest in Shares and performance rights
Shares: 5,500
Performance Rights: –

SUSAN (SUE) SMITH
Non-Executive Director, Dipl ClinRisk
Appointed 23 September 2019
Background
Mrs Smith manages an established consultancy business, providing advisory services to a range of healthcare organisations, investors and boards of directors. Mrs Smith has also hadled a distinguished career, serving for 14 years as Chief Executive Officer of The Princess Grace Hospital, London, and 11 years as the Chief Executive Officer of The Portland Hospital for Women and Children, London. Mrs Smith’s specific expertise is in the implementation of operational strategies within complex and acute care environments, and in the interaction with healthcare authorities and UK regulators. Her most recent role was as the Chief Executive Officer of the Independent Doctors Federation, a membership organisation representing practicing physicians within the UK independent healthcare sector.
Her past experience is now successfully translating into a diverse portfolio with non-executive director appointments having been successful in completing the Financial Times Non-Executive Director Advanced Professional Diploma. She is Board Chair of the Evewell (Harley St) Ltd, a fully integrated centre of medical excellence dedicated to caring for and protecting all aspects of fertility and gynaecological health. She also sits on an Advisory Board for Sweettree Home Care Services providing the bridge between hospital and community care. In the face of the ever-changing healthcare market Mrs Smith fosters first class relationships with a wide range of healthcare stakeholders to build first class services for patients.
Relevant Skills
- executive healthcare management
- leadership and strategy setting in complex environments
- risk management and governance
- customer relations
Committee Membership
Member of the Remuneration Committee
Member of the Nomination Committee
Current Directorships and other interests
Non-Executive Board Chair of the Evewell (Harley St) Ltd
Non-Executive Director of Elite Medicine Ltd
Trustee of the HCA International Foundation
Other listed company Directorships (last 3 years)
None
Relevant interest in Shares and performance rights
Shares: –
Performance Rights: –
JEFFREY ROSENFELD AC, OBE
Non-Executive Director
Appointed 29 January 2018
Background
Prof Rosenfeld is an internationally recognised neurosurgeon with extensive experience in senior healthcare medical and research executive roles and a distinguished and decorated career in the Australian Army. He is a retired Major General and a former Surgeon General, Australian Defence Force-Reserves. He has served on eight deployments to Rwanda, Iraq, Solomon Islands, Bougainville and East Timor. He was the Founding Director of Monash University Institute of Medical Engineering (MIME-Melbourne).

He is developing a bionic vision device to restore vision in blind people and he is also a leader in brain injury research. Prof Rosenfeld was Director of Neurosurgery at the Alfred Hospital for fifteen years, concurrently holding Professor and Head of the Department of Surgery at Monash University, for nine years. Prof Rosenfeld is active in many community organisations and champions various charitable causes. Prof Rosenfeld is an active volunteer in the Australian-Aid funded Pacific Islands Project which transfers clinical skills and knowledge to healthcare professionals in Papua New Guinea, Fiji and the Solomon Islands.
In 2018, Prof Rosenfeld was awarded the Companion of the Order of Australia, which is Australia’s highest civilian honour, the Meritorious Service Medal of the United States of America in 2017 and Officer in the Order of the British Empire in 2013.
Relevant Skills
- lifetime experience in providing healthcare
- clinical research and development
- board and Committee oversight and governance
- leadership and management
Committee Membership
Member of the Audit and Risk Committee
Member of the Nomination Committee
Current Directorships and other interests
Director of Vision for TBI Ltd
Former Major General, Australian Defence Force (Army Reserve)
Other listed company Directorships (last 3 years)
None
Relevant interest in Shares and performance rights
Shares: 1,693
Performance Rights: –

STAN MCLIESH
Non-Executive Chair, B Ed
Appointed 12 September 2002, Ceased Directorship 30 November 2019
Background
Mr McLiesh has vast experience across pharmaceutical research and development, and distribution and commercialisation of pharmaceutical products. He was closely involved in the transition of CSL Limited (ASX: CSL) from government ownership through corporatisation to a highly successful listed company as General Manager. During this time, he helped CSL expand its international reach, brokering numerous in-licensing agreements, M&A transactions and partnerships with
multinational firms, becoming the most successful Australian life-sciences company. Mr McLiesh has previously served in non-executive roles in the medical device field. As Chair of CLINUVEL from 2010 to 2019, Mr McLiesh was involved in formulating the successful European commercial strategy for SCENESSE® (afamelanotide 16mg) and overseeing the continuity and stability of the CLINUVEL Group.
He has taken a leading role in setting US commercial strategy, culminating in US FDA’s approval of SCENESSE® in October 2019.
His ability to navigate through crises and oversee clear pathways towards finding solutions made him highly capable to steer management over many years, up until his retirement in November 2019.
Relevant Skills
- pharmaceutical research & development, commercialisation
- commercial acumen
- general management
- experienced in listed company Directorships
Committee Membership
Member of the Remuneration Committee
Member of the Audit and Risk Committee
Member of the Nomination Committee
Current Directorships and other interests
Vice President of the Board of Ivanhoe Girls
Grammar School, Melbourne
Relevant interest in Shares and performance rights
Shares: 187,774
Performance Rights: –
Other listed company Directorships (last 3 years)
None
INFORMATION ON COMPANY SECRETARY
DARREN KEAMY
Company Secretary, Chief Financial Officer
Qualifications: BComm, CPA, GradDip ACG
Mr Keamy, a Certified Practicing Accountant and Company Secretary, joined CLINUVEL in November 2005 and became Chief Financial Officer of the Group in 2006. He has previously worked in key management accounting and commercial roles in Amcor Limited and has experience working in Europe in financial regulation and control within the banking and retail pharmaceutical industries. He has overseen the financial management of the Group since 2005, played a role in raising AUD$95 million in capital, and assisted the steering of the Group from a loss-making, pre-revenue position to a commercially focussed profitable enterprise.
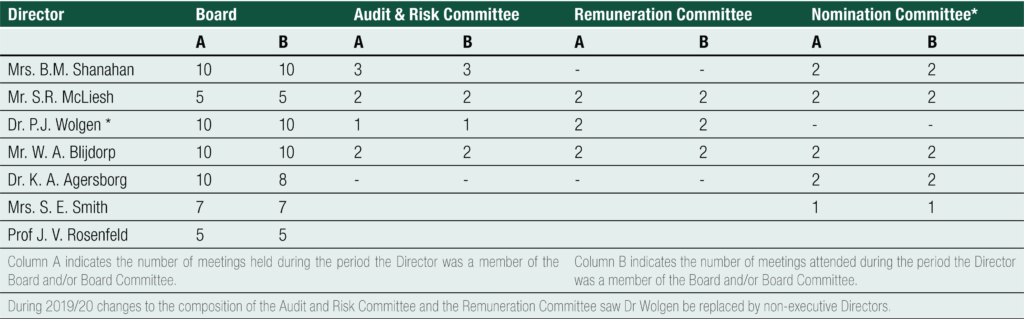
MEETING OF DIRECTORS
The following table summarises the number of and attendance at all meetings of Directors during the financial year:
PRINCIPAL OBJECTIVES AND ACTIVITIES
Objectives
CLINUVEL PHARMACEUTICALS LTD (CLINUVEL) is a global biopharmaceutical company focussed on developing and delivering treatments for patients with a range of genetic and vascular disorders. CLINUVEL’s pioneering work in melanocortins aims to translate scientific breakthroughs to innovative medical solutions for complex problems and thus deliver lifelong care and novel products to patients and consumers.
CLINUVEL’s expertise in understanding the interaction of light and human biology is focussed on preventing the symptoms of genetic diseases related to the exposure to the visible light spectrum and UV radiation along with addressing a range of depigmentation disorders. These patient groups range in size from 5,000 to 45 million worldwide.
CLINUVEL has developed and launched the world’s first systemic photoprotective drug in Europe and the USA. During the year, the scope of CLINUVEL’s research and development program was extended to the application of melanocortins to treat acute disorders and vascular anomalies.
The long-term financial objective of the Group is to maximise company value through the distribution of treatments to patients in need. The key to long term sustainable performance is:
- continuing the successful research and development of a portfolio of assets centred around its key drug candidate SCENESSE® and its melanocortin derivatives;
- the successful commercialisation, manufacture and distribution of these products; and
- maintaining financial discipline and stability.
Performance Indicators
Management and the Board monitor the overall performance of the Group in the achievement of its objectives in relation to a defined strategic plan and annual operating and financial budgets.
The Board, with Management, have identified a range of key performance indicators (KPIs) that are used annually to monitor performance. Key managers monitor performance against these KPIs and provide regular reports to the Board for review, feedback, and guidance, as necessary. This enables the Board to actively monitor and guide the Group’s performance.
Activities
The principal activities of the Group during the financial year were to:
- manage the commercial distribution in Europe of its leading drug product SCENESSE® (afamelanotide 16mg) for the treatment of a rare, genetic metabolic disorder, erythropoietic protoporphyria (EPP);
- establish commercial distribution of SCENESSE® in the USA following the approval of the US Food and Drug Administration (FDA) in October 2019 of SCENESSE® for the treatment of adult EPP patients;
- progress the ongoing research and development of its product pipeline for a range of severe disorders, including:
- SCENESSE® in combination with narrowband ultraviolet B (NB-UVB) phototherapy and topical pharmaceutical formulations of melanocortin analogues for the treatment of the skin depigmentation disorder, vitiligo;
- topical over-the-counter formulations for photoprotection of the skin;
- medicinal photoprotection through DNA repair of the skin; and
- the development of PRÉNUMBRA®, a new liquid formulation of afamelanotide for the treatment of critical indications to be announced.
REVIEW OF OPERATIONS AND FINANCIAL CONDITION
Key Features of Business Operations
There are several key features of CLINUVEL’s business operations:
- The commercial operations of the Group are undertaken in Europe and the USA.
- Since June 2016 CLINUVEL has distributed SCENESSE® to EPP patients through accredited Expert Centres, working within the commitments agreed with the European Medicines Agency (EMA) as a condition for continuous marketing authorisation.
- Since April 2020, CLINUVEL has been treating patients with EPP through accredited Specialty Centers in the US, in accordance with the approval of the FDA, granted in October 2019.
- The net price per unit of SCENESSE® is uniform across the jurisdictions in which it operates.
- Distribution costs specific to each jurisdiction determines the gross price of SCENESSE®.
- This reflects the Group’s values of fairness and equitable access to treatment by all patients.
- SCENESSE® is manufactured in the USA by a sole contract manufacturer and is distributed by the Group directly to accredited Expert Centres in Europe and Specialty Centers in the USA.
- CLINUVEL’s cash receipts are markedly higher in the northern hemisphere during spring and summer when ambient light is more intense and demand for treatment from EPP patients is higher than in autumn and winter.
- The Group has an ongoing clinical interest to further develop SCENESSE® and its derivatives with a focus on vitiligo, a skin depigmentation disorder; and DNA repair of the skin, in an undisclosed indication.
- The research and development program has been extended through the development of a second formulation of afamelanotide, PRÉNUMBRA®, with a focus on its application to acute disorders and vascular anomalies in indications to be announced.
- The Group’s product development program is conducted through its fully owned Singaporean subsidiary, VALLAURIX PTE LTD (VALLAURIX).
- The Melbourne headquarters of the Group covers the key regulatory affairs, scientific programme, finance, and investor relations functions, whilst the United Kingdom office co-ordinates global operations, communications, and marketing.
Review of Operations
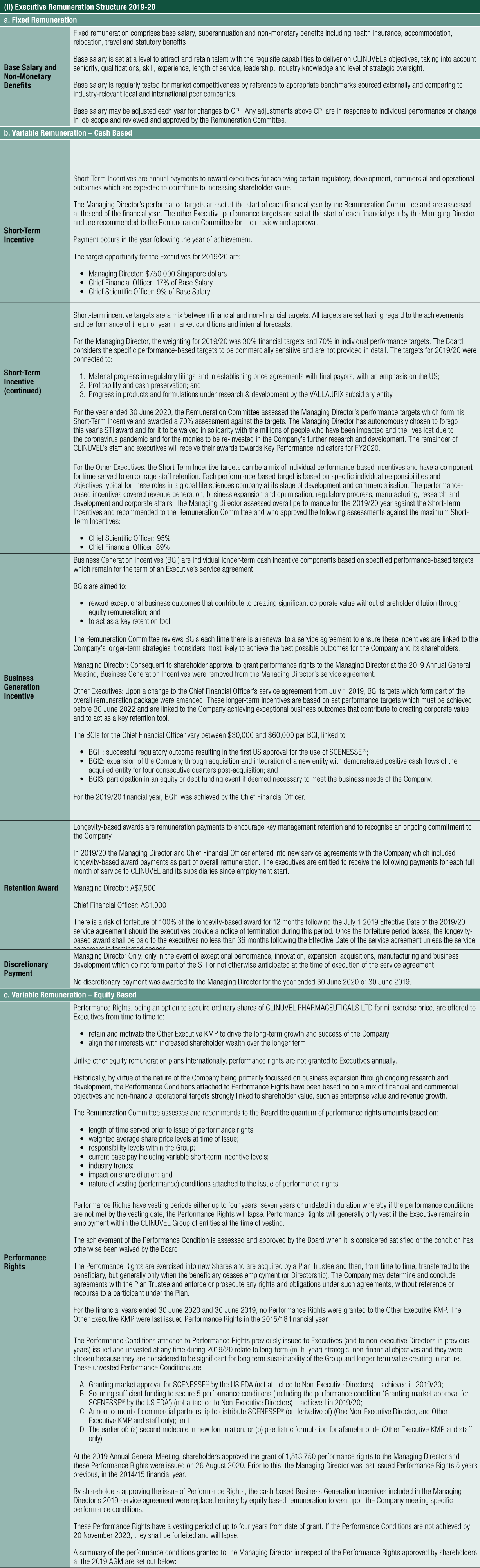
The review of operations for FY2020 focuses on the distribution of SCENESSE® in Europe and the USA, ongoing work to obtain regulatory approval of SCENESSE® in new jurisdictions, the expansion of the Group’s laboratory facilities in Singapore, and the progression of the product pipeline to develop SCENESSE® and its analogues for the treatment of patients with a range of severe genetic, skin, and vascular disorders.
Distribution of SCENESSE® in Europe
The supply of SCENESSE® to EPP Expert Centres across key European countries, including under a special access scheme to Switzerland, continued in the year ended 30 June 2020 (FY2020). During the corona-pandemic, the majority of EPP Expert Centres continued prescription of SCENESSE® due to the ongoing clinical demand, while a small number of Centres either deferred orders or reduced order sizes in the initial months of the COVID infections. These few Centres were not able to provide treatment access to patients, or patients were unable to travel to Centres. Despite the uncertainty surrounding the pandemic, patient demand for SCENESSE® remained high, with existing patients continuing to seek treatment and new patients receiving treatment for the first time.
We continue to progress reimbursement of the cost of treatment with authorities in other European countries.
Distribution of SCENESSE® in the USA
On 8 October 2019, the FDA approved SCENESSE® to increase pain free light exposure in adult patients with a history of phototoxic reactions from EPP. This was a milestone approval for the Group after 15 years of research and development of SCENESSE® for EPP which had an unmet medical need for treatment. Following the FDA’s approval, the Group activated its implementation plan for US operations and within six months of approval, completed the key pre-distribution logistics to commence treatment. These logistics included establishing the business infrastructure, identification of the correct codes for treatment to ensure smooth operations and reimbursement, initial insurer discussions and agreement to reimburse the cost of treatment, and identification of the initial Specialty Centers to be accredited and trained by CLINUVEL.
In April 2020, CLINUVEL commenced distribution of SCENESSE® for adult EPP patients with the first US insurance companies initiating reimbursement for treatment under Prior Authorization (PA). Over 40 insurance companies have now agreed to consider SCENESSE® under PA. CLINUVEL has established a Savings Program to assist with the out-of-pocket expenses of patients and provides a dedicated patient and healthcare professional website to facilitate patient access to treatment. CLINUVEL actively supports patients and Specialty Centers in their applications to insurance companies for approval to reimburse the cost of treatment of SCENESSE®.
Our plan is to accredit 30 Specialty Centers over a phased period. At the time of writing, 17 Specialty Centers have been accredited, which is ahead of our planning. Cash receipts for the financial year ending 30 June 2020 did not include any receipts from the supply of SCENESSE® in the US market. The Company expects, in these early stages of US launch, that payment terms may be longer in duration than the 30 to 60 days average length of payment term in Europe. Modest revenue was recorded in the first few months of treatment to 30 June 2020 and the outlook for the US business is underpinned by the progress being achieved in the number of Specialty Centers accredited and patients treated.
SCENESSE® for EPP in New Jurisdictions
With regulatory approvals from the EMA in Europe and more recently, the FDA in the USA, and information on the patient experience in Europe generated from its post-marketing commitments, the Group continues to work towards gaining regulatory approval for SCENESSE® for EPP patients in other important markets. This reflects our commitment to provide EPP patients worldwide with access to SCENESSE®.
In October 2019, the Australian Therapeutic Goods Administration (TGA) granted SCENESSE® the right to be filed under its priority registration process. In December 2019, CLINUVEL applied to the TGA for SCENESSE® to be registered in the Australian Register of Therapeutic Goods (ARTG). If registered, SCENESSE® would be made available by prescription in Australia for the prevention of phototoxicity in adult patients with EPP. In January 2020, the TGA accepted the registration dossier for review. A decision is expected during the fourth quarter of calendar 2020. In parallel, interactions with the Pharmaceutical Benefit Scheme (PBS) have occurred to exchange information on risk, benefit, and budget impact in Australia. The aim is to assess whether SCENESSE® can become listed on the PBS in Australia. It is expected that the drug will be made available exclusively through outpatient departments of speciality centres since it will be administered by specialists only.
In April 2020, the Group commenced a Collaboration Agreement to launch SCENESSE® (afamelanotide 16mg) under a Named Patient Program (NPP) for the treatment of EPP patients in the People’s Republic of China. The collaboration with HK Winhealth Pharma Group Co. Limited (Winhealth) focuses on facilitating early access for Chinese EPP patients while collecting data for a new drug application (NDA) to the Chinese National Medical Products Administration (NMPA). CLINUVEL and Winhealth will work with prominent hospitals in China to facilitate EPP patient treatment. The NPP will include up to 10 Chinese EPP patients – treated according to US and EU protocols – who will be evaluated during a defined period. Local subsidies are available to enable eligible EPP patients to receive treatment. Following treatment with SCENESSE® under the NPP, CLINUVEL and Winhealth will evaluate the safety and effectiveness in Chinese EPP patients. The collaboration will also focus on subsequent registration of SCENESSE® on the National Drug Reimbursement List. On a prevalence basis, an estimated 5,000 Chinese residents suffer from EPP, for which there is no approved therapy.
An application was also lodged during the year for regulatory approval to distribute SCENESSE® in a non-EU country, and submissions to regulatory authorities in Japan and Latin America are planned.
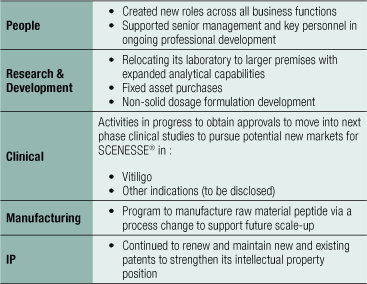
Expansion Singapore Laboratory
During FY2020, CLINUVEL invested in the further expansion of its facilities in Singapore with new state of the art and expanded laboratories to further progress R&D on novel melanocortins, and prescription and over-the-counter products. In February 2020, the Group announced that the research and development capacity of its wholly owned subsidiary, VALLAURIX, will be expanded through both a new biological and analytical laboratory, which are planned to work according to both ISO17025 and Good Laboratory Practice (GLP) specifications. CLINUVEL has added new highly skilled local personnel to its existing team and specialised technical laboratory equipment to further enhance the progress of its product pipeline. VALLAURIX has received support of its expansion plan from the Singapore Economic Development Board (EDB) with an award under their Research Incentive Scheme for Companies (RISC). This is part of the Government of Singapore’s incentives to assist Singaporean businesses to develop their research capacity to advance high valued technologies. The EDB award is up to S$500,000 (A$547,000) over 3 years. The opening of the laboratory was planned for July 2020, but due to the introduction of prudent regulations by the Singapore Government to contain the corona-pandemic, it is expected the new facilities will be completed by the end of the third quarter of calendar year 2020.
Product Pipeline
The Group has an extensive product development pipeline that encompasses the application of SCENESSE® and other novel treatments for patients with severe genetic, skin, and vascular disorders which lack therapeutic alternatives.
The pipeline includes research and development into:
- SCENESSE® for adult vitiligo patients;
- next generation products based on melanocortin analogues CUV9900 and VLRX001, currently being evaluated as an adjuvant maintenance therapy in vitiligo, with the intention of developing these analogues for medicinal purposes to be administered topically;
- a range of over-the-counter products for general photoprotective application;
- the use of melanocortins in DNA repair of the skin; and
- the application of a newly developed second formulation of afamelanotide, PRÉNUMBRA®, a liquid controlled-release formulation, to be evaluated in clinical trials for acute disorders and vascular anomalies.
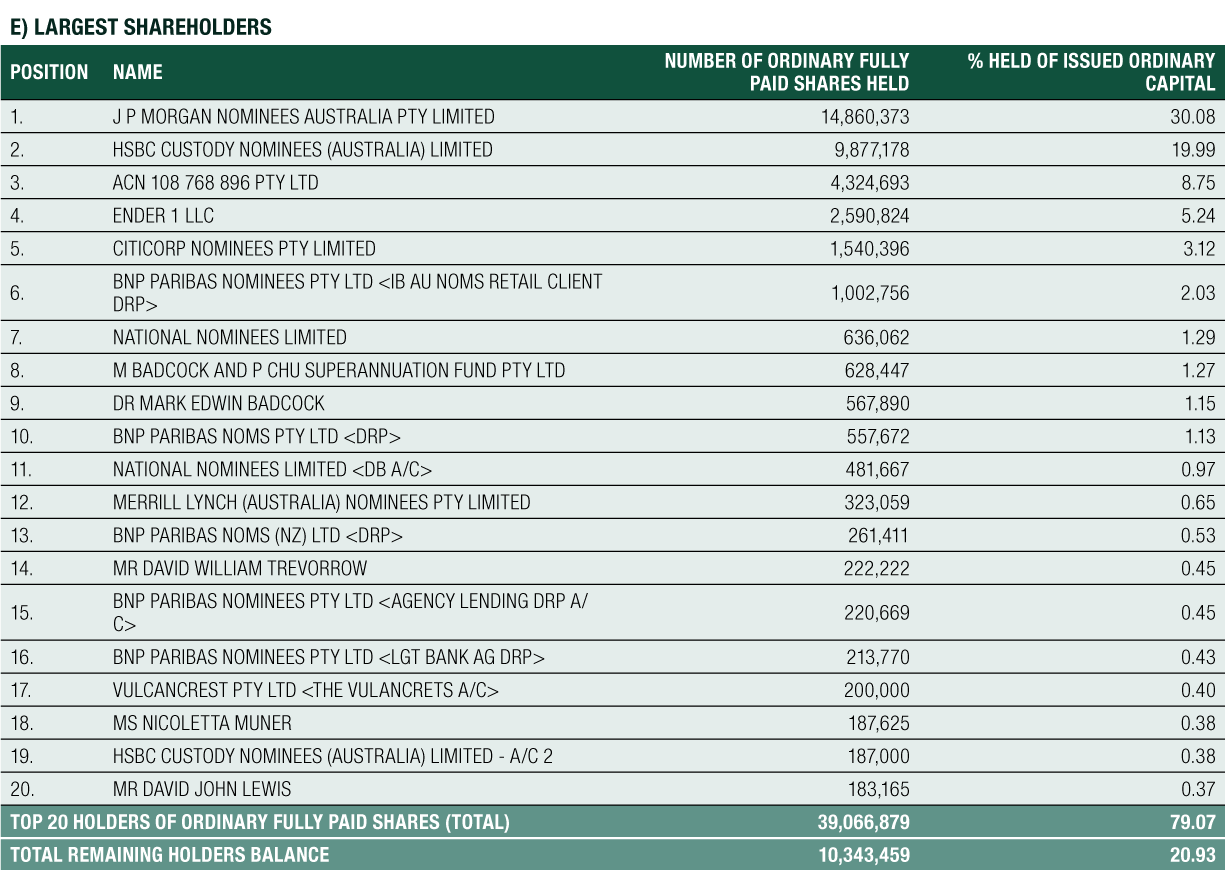
Financial Review
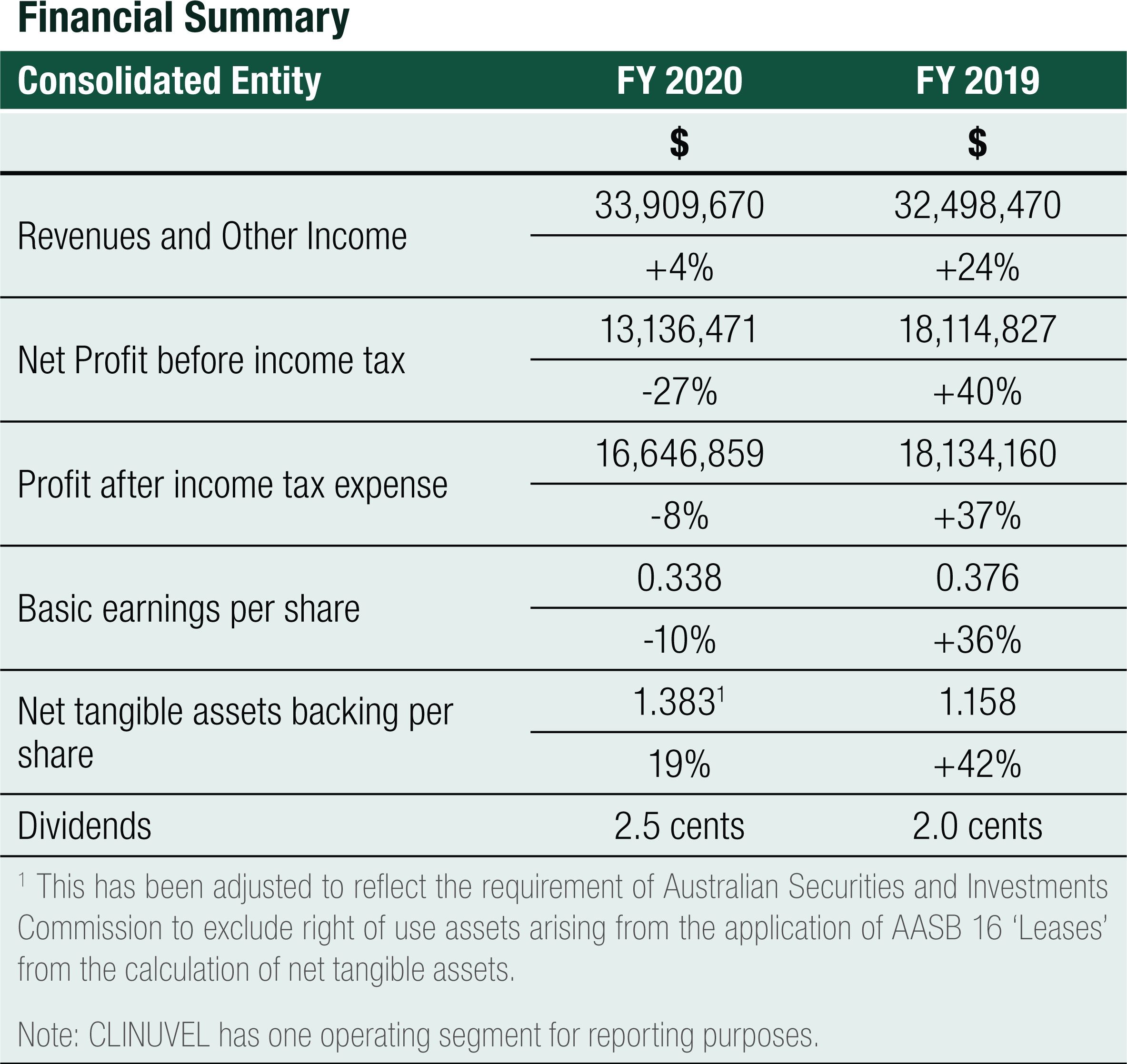
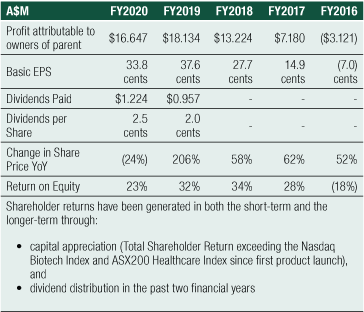
The financial year ended 30 June 2020 marks the completion of the Group’s fourth consecutive year of achieving a net profit, a positive cash flow result and increased revenue growth.
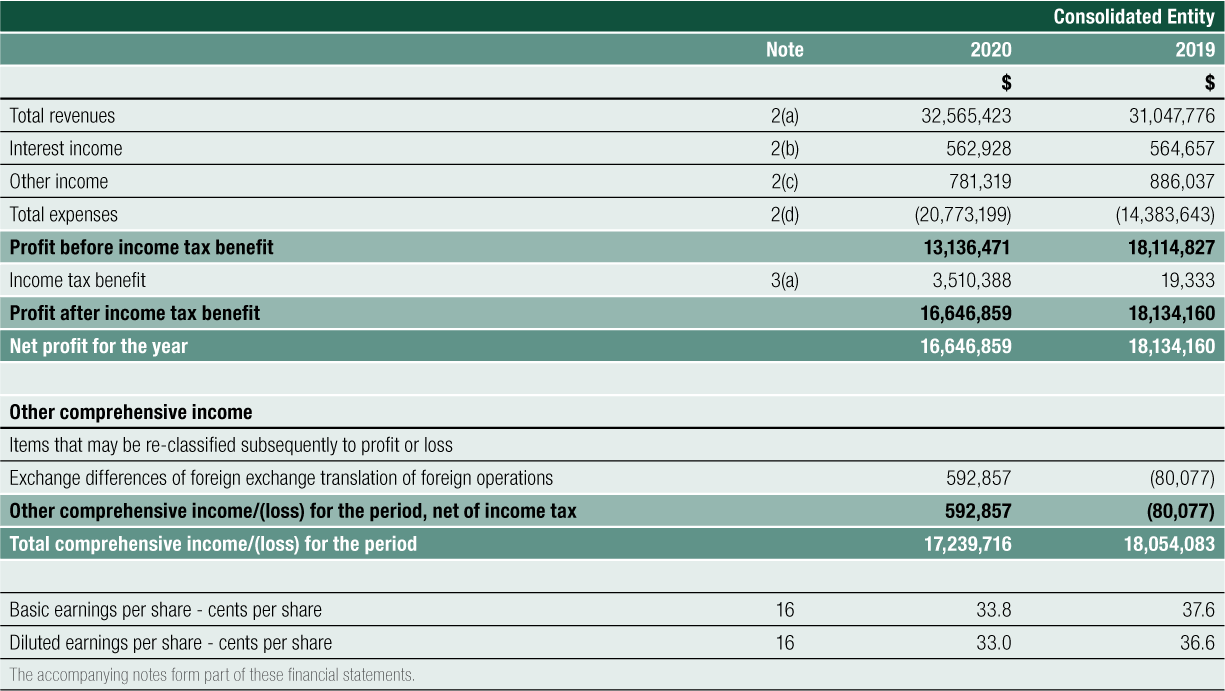
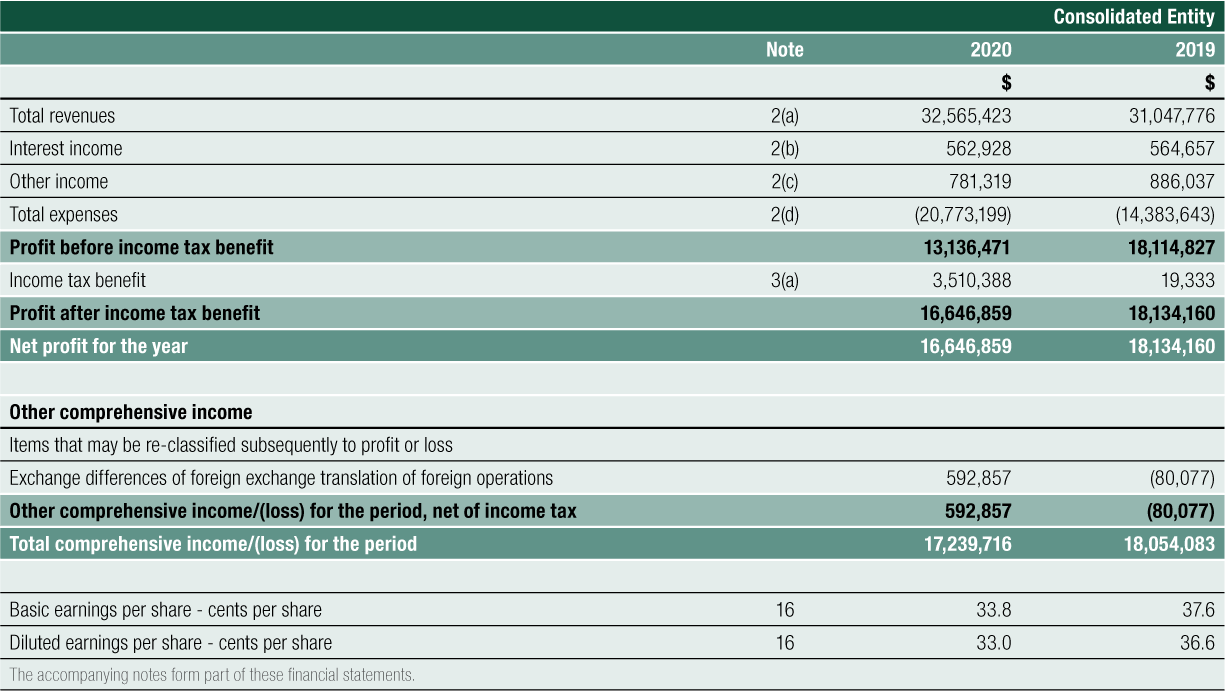
The result for the Group for FY2020 was a $13.136 million profit before tax, compared to $18.115 million for FY2019, a 27% decrease. The result reinforces the Group’s primary strategic focus to grow its commercial operations of SCENESSE® in the EU and the US and to prepare for future product growth and business expansion. Total expenses increased by 44% year-on-year, complemented by a combined increase in total revenues, interest income and other income of 4% year-on-year.
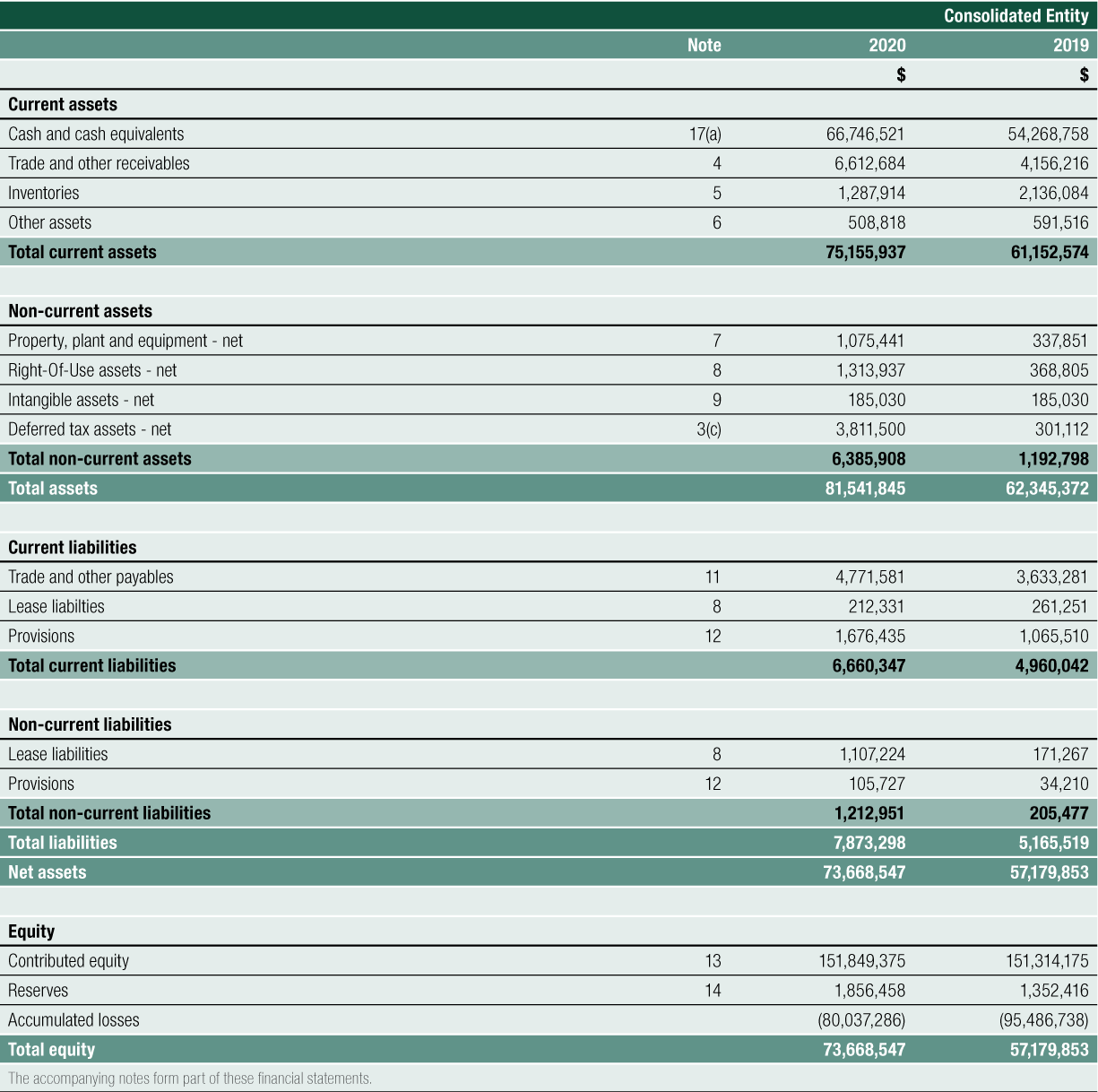
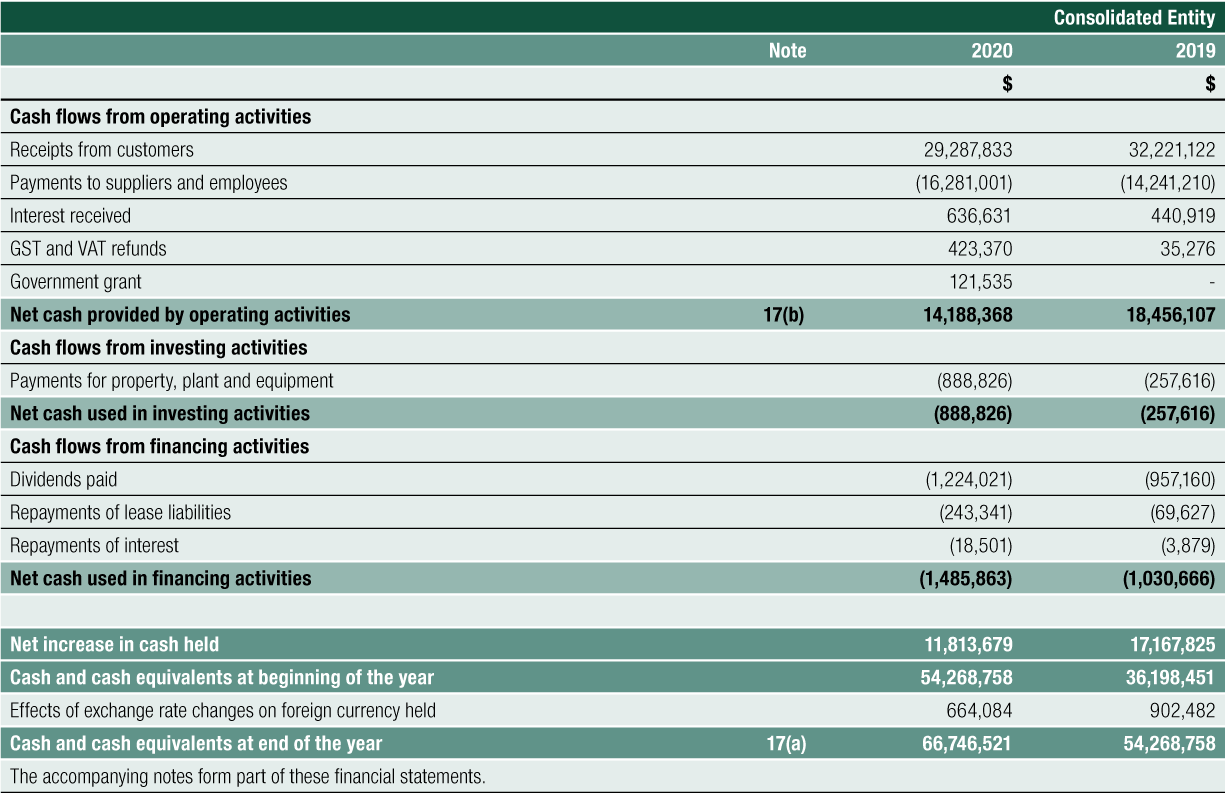
Net Cash provided by Operating Activities was $14.188 million for FY2020. After the deployment of cash in investing and financing activities, net cash added $12.478 million to cash and cash equivalents on the balance sheet. Cash reserves have increased steadily since 2016, from $14.170 million to the 30 June 2020 level of $66.747 million, a 44% compound annual growth rate over the last 4 years
Financial Summary
Revenues
The Group achieved Total Revenues of $32.565 million for FY2020, a 5% increase on the prior year revenue result of $31.048 million.
A comparison of the FY2020 reported and constant currency results against the FY2019 reported results for Commercial Sales and Special Access Scheme Reimbursements is shown below:
Commercial Sales - Europe
On a constant currency basis commercial sales revenues of SCENESSE® in Europe decreased 7.9% for the year. The result was driven by a combination of:
- EPP Expert Centres located in regions severely affected by COVID19 either deferring or reducing orders at a time when orders typically increase in the warmer months;
- EPP Expert Centres running down inventories, offset by
- Further increases in new patients enrolled under the post-authorisation safety setting and treated by EPP Expert Centres before and during lockdown.
Commercial Sales - USA
First commercial sales occurred in the latter part of FY2020 to one EPP treatment centre. Reimbursement of SCENESSE® was initiated by first-mover US insurance companies for treatment under Prior Authorization (‘PA’) arrangements. More than forty insurers have agreed to reimburse SCENESSE® either via PA or through acceptance of the drug on individual formularies.
Reimbursements – Special Access Schemes
The distribution of SCENESSE® under Special Access Schemes continued to provide a preventative treatment for adult EPP patients primarily to Switzerland. SCENESSE® was also exceptionally supplied outside Switzerland under a special access arrangement whereby CLINUVEL received full cost compensation, linked to the uniform price of SCENESSE® sold in Europe under the marketing authorisation.
On a constant currency basis, sales reimbursements from special access schemes increased 29.9% for the year. The result was driven by:
- Growth in the average number of presciptions per patient throughout the course of the year, and
- New patients receiving treatment for the first time
Other Income
Interest Revenue and Other Income
This exposure in holding funds in non-Australian dollar currency, combined with revaluing end date trade debtors and creditors from their original currency into Australian dollar presentation currency, contributed to the Group reporting a gain of $0.537 million for FY2020 (FY2019: $0.886 million gain).
The Group recorded other income of $0.127 million in government grants received in Australia and Singapore to assist companies responding to the economic impact of the COVID19 pandemic. The Group also benefited from realising exchange rate gains on transactions in non-Australian currency throughout the year of $0.117 million.
Interest received from funds held in bank accounts and term deposits for the year ended 30 June 2020 was $0.563 million compared to $0.565 million for year ended 30 June 2019.
The positive financial performance of the Group saw an increase over the 12 months to 30 June of $12.478 million to its cash reserves. Over the course of FY2020 the Company was able to transfer more funds into higher-yielding Australian dollar fixed-rate term deposits.
The average amount of cash held in term deposits was 55% higher than for FY2019. However, the higher cash balances were offset by a lower interest rate yield it earned on holding interest-bearing term deposits, averaging 95 basis points less year-on-year. The decrease in interest rate yield reflected the impact of Australian government monetary policy on term deposit rates on offer throughout the year. The Group’s policy to maintain lower-yielding foreign currencies to cover working capital requirements is reflected in this result. Funds held in non-Australian dollar currency providing a natural hedge against downward movement on the Australian dollar. The average amount of funds held in non-Australian dollar currency in FY 2020 has remained stable, decreasing 4% on average when compared to FY2019.
Expenditures
Total Expenses for the Group for FY2020 were $20.773 million. There was a deliberate and controlled increase in expenses of 44% during the year compared to FY2019 to support the Company’s strategic initiatives, including investment in the research and development program for future organic growth.
The Group maintained its focus on its expenditure mix as it has done throughout the SCENESSE® development program. Overall, total R&D and commercialisation expenditures accounted for 46% of the Group’s total expense result for FY2020, compared to 48% for FY2019. Whilst the expenditure mix showed a 2% decline, the total expenditures on R&D and commercialisation costs, comprising clinical study costs, drug formulation research, manufacture and distribution, regulatory fees and research, development and commercialisation-specific overheads such as personnel, were $9.630 million in FY2020, increasing 40% from $6.871 million in FY2019. The increase in these overall expenditures reflects the Group’s focus throughout the year to further invest in its commercial rollout to secure revenues in the EU and for the first-time, the USA.
Clinical Development
Clinical development fees increased 102% from $0.091 million in FY2019 to $0.185 million in FY2020.
Since the granting of market authorisation by the EMA in late 2014, the Group has prioritised its commercialisation activities in the EU and in pursuing a regulatory approval in the USA ahead of advancing its clinical trial program. This has been reflected in expenses towards clinical development representing approximately 1% of total expenses in each year since the year of European regulatory approval. Moving forward, the Group intends to invest funds in its clinical activities, both in the use of SCENESSE® in various therapeutic fields and in the clinical development and testing activities of the new products and formulations as part of the VALLAURIX operations.
This expense result for FY2020 was driven by:
- Increased clinical expert support services to advise on initiatives on the expanded use of SCENESSE®, and
- Growth in product development and testing services in the VALLAURIX operations under laboratory setting.
Drug Formulation R&D, Manufacture & Distribution
Expenses toward further research, development, manufacture and optimisation of the implant drug formulation and the freighting and distribution to the end user increased 52%, from $2.388 million in FY2019 to $3.624 million in FY2020.
The Group continues to invest in its manufacturing supply chain to prepare for future sales growth and to meet short-term and long-term inventory requirements. During the year the Group embarked on a manufacturing program to replenish raw material peptide via a process change to support future scale-up. Validation of the process change is underway and will extend into FY2021. Continuous process improvement initiatives with the implant contract manufacturer were a part of the batch manufacturing campaigns that were conducted during FY2020. New distribution centres with contracted parties and third-party service providers were established in Europe to respond to the UK’s pending departure from the EU as part of Brexit and to support supply to US EPP Expert Centres. Drug formulation R&D also includes the development work and usage of derivative peptide material within the VALLAURIX Singapore operations.
This expense result for FY2020 was driven by:
- Cost increases passed to the Group from its contract manufacturer as part of FY2020 implant manufacturing campaigns;
- Investment in process development of afamelanotide raw material peptide manufacturing which had commenced in FY2020;
- Growth in handling and freighting activities to support the movement of serialised goods around Europe under the Falsified Medicines Directive and to move goods in the US; and
- Increased formulation development work in topical formulations, recognising the usage of derivative peptides.
Clinical, Regulatory & Commercial (C,R&C) Overheads
C,R&C overheads increased 32% from $2.948 million in FY2019 to $3.893 million in FY2020.
As part of CLINUVEL’s longer term objectives, increasing the C,R&C personnel headcount is considered an essential investment to:
- drive the new product development program in its internal innovation centre, VALLAURIX PTE LTD;
- establish and grow a commercial distribution program in the USA;
- further sustain the ongoing commercial activities in Europe; and
- investigate the further use of SCENESSE® in indications other than EPP for the aim to expand its market potential.
- Increased global staff headcount across its scientific affairs and commercial affairs teams.
Regulatory (Pre- & Post-Marketing) & Non-clinical
Regulatory and non-clinical fees increased 33% from $1.444 million in FY2019 to $1.928 million in FY2020.
Fees related to regulatory affairs for both pre- and post-marketing activities are directly related to the Group’s strategic focus in the current year to meet its ongoing regulatory compliance activities to distribute SCENESSE® in Europe and in the USA. These activities include pharmacovigilance, safety reporting, PASS registry data capture and dossier updates. SCENESSE® is established as standard of care in a number of European countries. The first treatment results from the European EPP Disease Registry study were independently published during FY2020, showing ongoing longer-term maintenance of the safety profile of SCENESSE® and clinical benefit for patients receiving treatment.
Pre-marketing regulatory fees for FY2020 included finalising and submitting an application to the Australian Therapeutic Goods Administration for the use of SCENESSE® in EPP. An outcome is anticipated in late 2020.
Regulatory and non-clinical fees also include the support required for pricing dossier submissions and in responding to the pricing negotiations.
This expense result for FY2020 was driven by:
- Growth in ongoing pharmacovigilance services; and
- Fees to prepare and respond to an increased number of audits and inspections from various regulatory bodies including the UK MHRA and FDA.
Business Marketing & Listing
Business marketing and listing fees increased 26% from $1.502 million in FY2019 to $1.889 million in FY2020.
The Group has been further investing in its marketing and brand-building resources throughout the year. The focus to this business function has been to build a greater online awareness of the CLINUVEL brand in consideration of the US FDA’s approval of SCENESSE®. In addition, as the product development program in VALLAURIX progresses, resources and stragies are being put in place to support future product launch. Brand-awareness strategies were implemented during industry conferences at the start of FY2020 but were later impacted by the onset of COVID19.
- Additional in-house marketing resources through more personnel to support the building of CLINUVEL’s brand exposure across multiple forums and the promotion of VALLAURIX’s over the counter (OTC) products leading up to launch; and
- Increases to listing, share registry and corporate regulatory fees tied to market capitalisation growth and investor mix.
Patents and Trademarks
Patent fees increased 69% from $0.305 million in FY2019 to $0.516 million in FY2020.
Incurring expenditures in patent and trademarks provides the Group with essential protection and a competitive advantage over others.
This expense result was driven by:
- Further fortification of the intellectual property position on the new product development and complementary formulations within the VALLAURIX business;
- Further maintaining, strengthening and validating the position of the existing patent portfolio, including patent term extension requests; and
- Investments in trademarks of new product names including PRÉNUMBRA®, CLINUVEL’s new non-solid (liquid) presentation of its drug afamelanotide.
General Operations (incl Board)
Expenditures from general operations increased 61% from $4.923 million in FY2019 to $7.963 million in FY2020. General operations are reflective of the support function necessary to ensure the execution of the Company’s demanding near-term and long-term expansion strategy. Personnel costs including the remuneration of senior management is considered part of general operations along with IT, corporate support, legal, Board and various non-cash items.
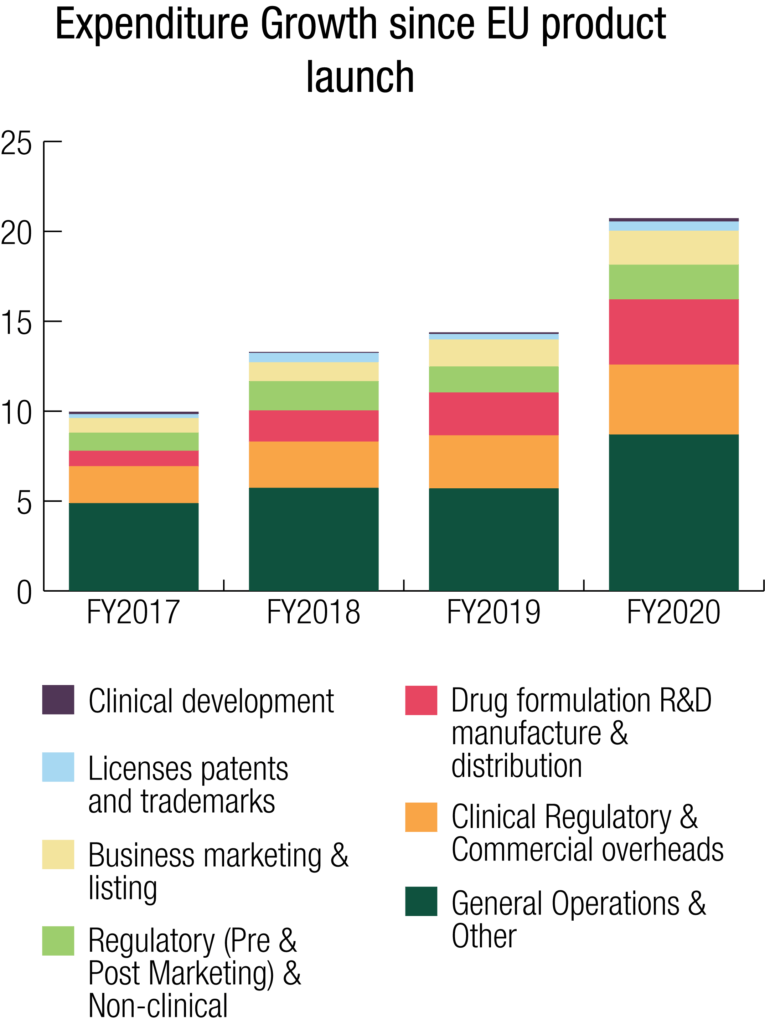
This expense result for FY2020 was driven by:
- Growth in non-cash expensing of share-based payments, the accounting charge directly related to the approval by shareholders of performance rights to the Managing Director at the 2019 AGM;
- changes to the remuneration arrangements of key management personnel, impacting salary and employee benefit expenses; and
- General increases to recruitment and insurances to support business growth, offset by savings to legal-related fees, principally from FY2019 including legal fees in connection to matters related to the EMA marketing authorisation and in responding to negotiations with England’s National Institute for Health and Care Excellence (NICE).
Other Expenses
Other expenses decreased less than 1% from $0.755 million in FY2019 to $0.750 million in FY2020.
Other expenses include travel and ad-hoc staff-related expenses. Whilst there was a steep increase in travel related costs in the first half of FY2020, staff mobility was severely restricted in the second half of FY2020.
Deferred Tax Asset
The Group has brought to account a deferred tax asset (DTA) relating to previously unrecognised prior period tax losses. resulting in a credit to income tax benefit of $3.510 million (FY2019: $0.019 million). The amount of the DTA brought to account reflects:
- the benefit to be received from utilising unused tax losses against the temporary differences that result in a deferred tax liability for the business; and
- the expected utilisation of unused tax losses against probable near term taxable profits.
Balance Sheet
One of the key objectives of the Company is to ensure its Balance Sheet is sufficiently positioned and robust enough to allow investment in future performance with a financial buffer to respond to unexpected adverse events. The Company has continued to preserve cash and cash equivalents held and, in doing so, is able to withstand anticipated increases to short-term liabilities to support the growth of the business and to sudden adverse economic conditions following unexpected events such as the coronavirus pandemic. This has been a deliberate and planned strategy, reflecting CLINUVEL’s conservative approach to risk management.
Key Balance Sheet highlights of the year:

The changes to the Balance Sheet was generated from positive cashflows flowing into the company from its commercial distribution program in the EU, increasing cash reserves by 23% from $54.269 million at FY2019 to $66.747 million at FY2020. Into FY2021, cashflows are expected to be received from the distribution of SCENESSE® in the US.
Total liabilities increased 52%, from $5.166 million to $7.873 million, with no long-term debt. The ratio of the Company’s overall debt to equity is 12%.
Shareholder Returns
Shareholder returns for FY2020 remain strong and are summarised (see first table right).
Investments for Future Performance
The Group’s key objectives are to progress CLINUVEL as a world leader in medicinal photoprotection and repigmentation and to support the expansion into other, similar genetic and skin-related disorders, as well as acute disorders and vascular anomalies. In addition to the ongoing development of its active and expanded product pipeline, the Group is open to consider the integration of new functions and capabilities through one or more selective acquisitions.
The Group has deployed working capital throughout the year to prepare for future performance across the following areas (see second table on right).
Capital Structure
The Group is debt free and has a sound capital structure of ordinary shares on issue plus unlisted securities in the form of conditional performance rights.
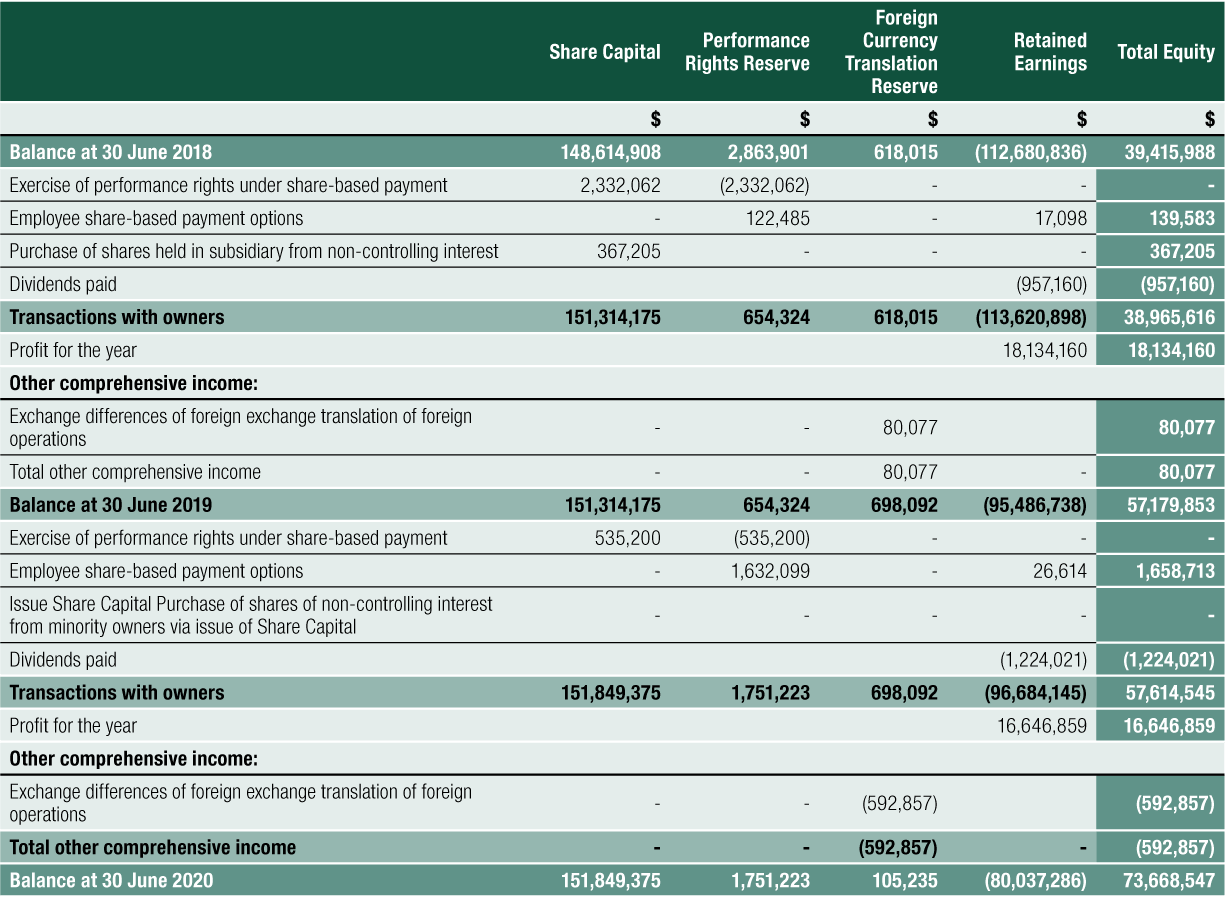
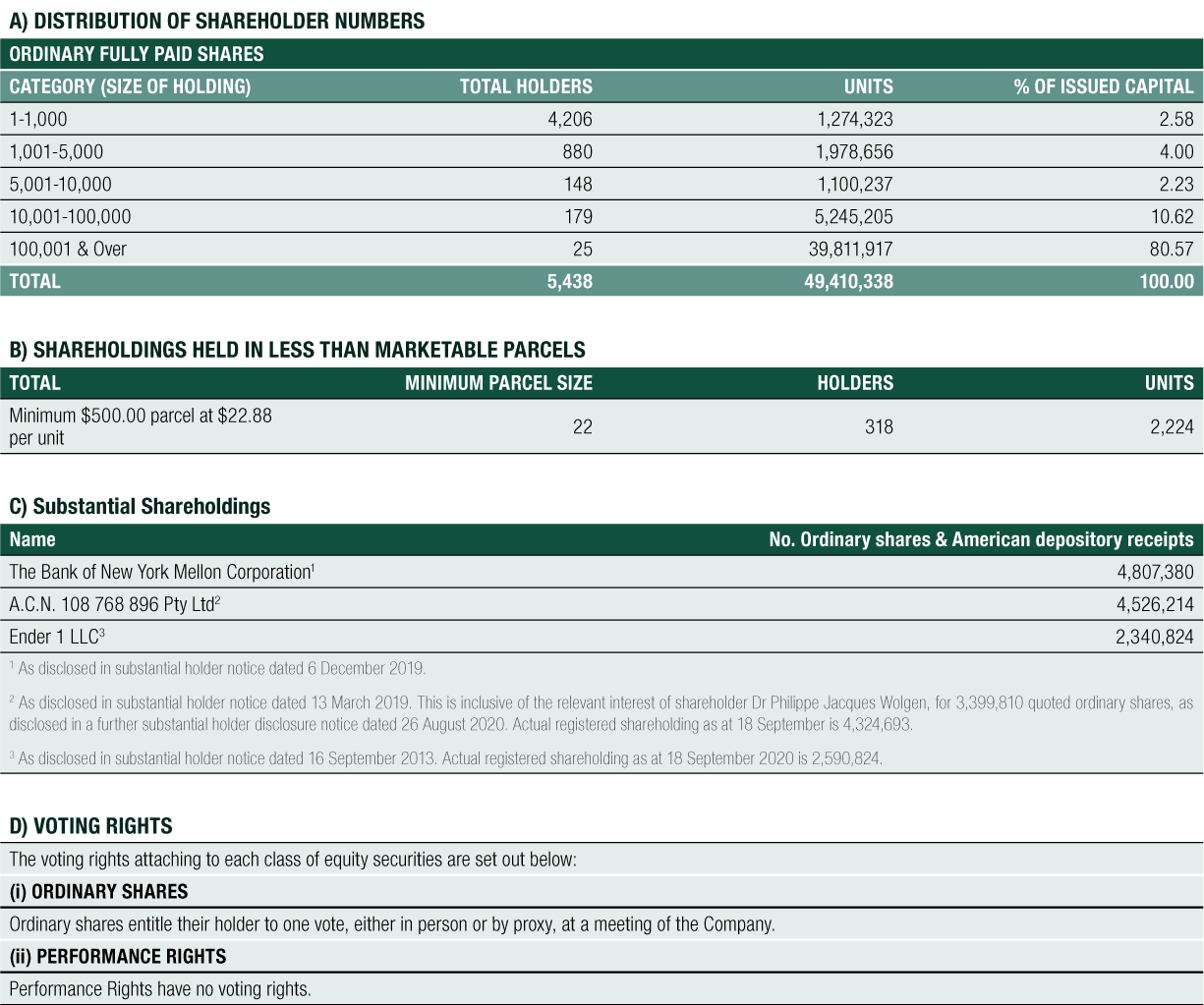
CLINUVEL’s outstanding shares on issue increased to 49,410,338 shares to 30 June 2020. The increase of 449,705 issued shares was through the exercise of performance rights under the Group’s performance rights plans.
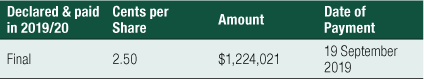
Dividends Paid or Recommended
Dividends paid or declared by the Group to members since the end of the previous financial year were (see right).
On 26 August 2020, the Board of Directors declared an unfranked dividend of $0.025 per ordinary share in relation to the full year ended 30 June 2020.
Cash from Operations and Other Sources of Cash
Overall, the Company generated $14.188 million in cash from its operating activities in FY2020 (FY2019: $18.456 million)
Cash inflows from customer receipts decreased 9% to $29.288 million compared to $32.221 million for FY2019.
Cash outflows from operations increased by 14%, from $14.241 million to $16.281 million.
There were also cash outflows of $0.889 million for the acquisition of property, plant and equipment, $0.262 million of repayment of borrowing and leasing liabilities and $1.224 million for the payment of an unfranked dividend to shareholders in relation to FY2019.
The Group’s policy towards cash management is to:
- Hold cash in at-call bank accounts and place additional cash in short-term term deposits providing favourable rates of interest; and
- Actively manage foreign currency exposure, taking account of recent and expected currency trends, holding foreign currencies as a natural hedge, using foreign exchange forward contracts and other foreign exchange risk management products, as considered appropriate.
The Group’s financial liquidity as at 30 June 2020 is reflected in:
- A quick ratio of 11.0:1 (30 June 2019 11.8:1); and
- Cash and cash equivalents of $66.747 million, accounting for 88.8% of total current assets (FY2019: $54.269 million, 88.7%of total current assets).
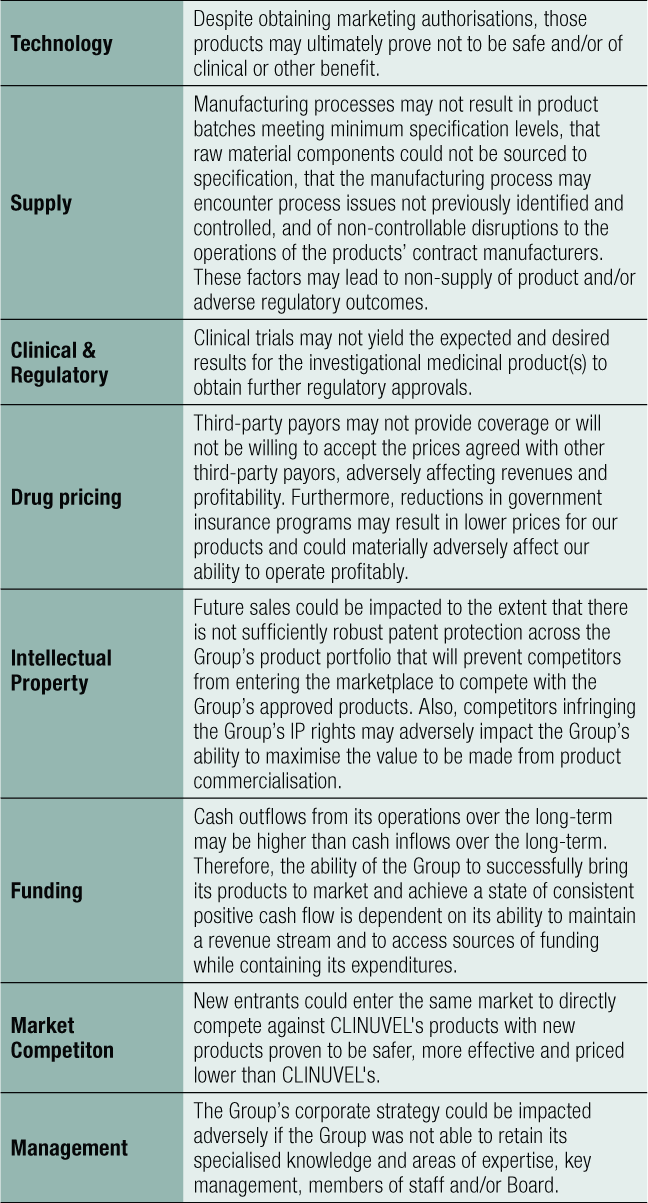
Material Business Risks
The following specific business risks are reviewed continually by the Board and Management, as they have the potential to affect the Group’s achievement of the business goals detailed above. This list is not exhaustive (see table below).
Impact of the Coronavirus Pandemic on CLINUVEL’s Business
The impact of the coronavirus pandemic on the human population across the globe is significant and has caused the most severe contraction in the world economy since the 1929 Great Depression. The impact and consequences on how we live, work, and interact will be felt for years. CLINUVEL is no exception to being impacted by the coronavirus pandemic. CLINUVEL’s business has proven resilient and is relatively well positioned to manage the difficult operating environment and progress its strategic initiatives.
Demand for SCENESSE®
Access to patients was affected particularly during the initial months when population lockdowns across Europe were first instituted. EPP Expert Centres either deferred orders or reduced order sizes in the initial months of the COVID infections because they were not able to provide treatment access to patients, or patients were unable to travel to them. Notwithstanding the uncertainty surrounding the pandemic, patient demand for SCENESSE® in Europe remained strong, with existing patients continuing to seek treatment and new patients receiving treatment for the first time. CLINUVEL is conscious of the patients it serves and the anxiety and uncertainty they face during the coronavirus-pandemic and it has worked to continue to meet their demand for SCENESSE®.
Research and Development
CLINUVEL’s research and development program continued to progress in FY2020. The operations of the laboratory facilities in Singapore were restricted during the circuit-breaker period, with some remote working required. The circuit-breaker also resulted in minor delays to the laboratory expansion project, now set to be completed by the end of the third quarter of calendar year 2020.
Supply of SCENESSE®
The sourcing, manufacturing and controlled distribution of SCENESSE® continued without material disruption or delay from the coronavirus pandemic. Raw material sourcing, manufacturing activities and movement of goods were able to be conducted without adversely impacting timeframes. CLINUVEL continuously reviews its operations to assess ongoing supply of SCENESSE® which may be impacted by the coronavirus pandemic.
CLINUVEL’s People
CLINUVEL has played a responsible role to assist the global effort to manage the spread of COVID-19. CLINUVEL personnel have adapted to work remotely, attending the office only as necessary and when permitted under government regulations. Video-based communications technology has been maximised whilst local and international travel has been minimised. Diligence under a difficult operating environment by the entire CLINUVEL team has seen productivity and focus remain largely unaffected.
Summary
CLINUVEL recorded a fourth consecutive annual positive cash flow and profit in FY2020. The impact of the corona-pandemic on the business during the second half of FY2020 has been managed and it has been able to respond to this challenge through the sound foundations established over a long period of time by management and the Board. CLINUVEL has entered FY2021 with cash reserves sufficient to respond to unforeseen negative global economic events.
Changes In The State Of Affairs
The Directors are not aware of any matter or circumstance not otherwise dealt with in this report that has significantly or may significantly affect the operations of the Group.
Significant Events After The Reporting Date
There has not been any matter, other than reference to the financial statements that has arisen since the end of the financial year that has affected or could significantly affect the operations of the Group, other than:
- On 26 August 2020, the Board of Directors declared an unfranked dividend of $0.025 per ordinary share.
Likely Developments And Expected Results
The Group launched SCENESSE® in Europe in June 2016. As part of the conditions attached to the European marketing authorisation, the Group operates an agreed long-term risk management plan under the supervision of the EMA. The Group has been assisted by third parties to support the European EPP Disease Registry to monitor long-term safety and it will continue to invest in existing and new personnel with the appropriate skills and expertise to maintain the ongoing requirements of the post-authorisation program in Europe. The ongoing requirements will remain in place until such time the EMA decides these are no longer necessary.
The Group has established a reference price for SCENESSE® as part of its uniform pricing strategy in Europe and has entered into pricing agreements with several European countries, and state and private insurance groups. The Group has established a distribution-focused workforce in Europe to support the increase in product volumes and will continue to increase staff numbers as more pricing agreements per country are established with payors, and as the required pharmacovigilance activities continue to expand.
The Group has focused on its manufacturing requirements by working with its contract manufacturer and raw material supplier to meet commercial product supply in line with its timing expectations and to pursue ongoing process improvement initiatives to support future increases in supply. These initiatives are part of continuous improvement and will form part of the Group’s expenditure base moving forward. The contract manufacturer bears responsibility for the manufacturing standards of the commercial drug product.
The US FDA approved SCENESSE® for the use in EPP during the financial year. SCENESSE® was launched in the US in April 2020. The Group is focussed on securing agreement on reimbursement of SCENESSE® with insurers to make SCENESSE® available to all US patients receptive to the treatment. The Group will continue to expand its resources and activities to support US market entry which includes operating a risk management plan similar to what has been instituted in Europe.
The Group will continue its North American clinical program to evaluate the effectiveness of its lead product to repigment vitiliginous lesions (depigmented skin areas) in combination with NB-UVB light therapy in patients with vitiligo. This program would include advancing into the next phases of clinical studies to demonstrate the efficacy and long-term safety of SCENESSE® in combination with NB-UVB in the treatment of vitiligo.
The Group also intends to further progress its clinical program with SCENESSE® in other indications, including yet to be disclosed acute and critical disorders. To support this likely development, CLINUVEL is advancing PRÉNUMBRA®, a non-solid dosage form of afamelanotide as a potent haemodynamic, vasoactive and anti-oncotic therapeutic agent, initially in adult patients.
The Group expects to advance its product pipeline, progressing the development of the molecules CUV9900 and VLRX001 through the various development phases which may include formulation development, non-clinical and human testing. In addition, complementary OTC products are being developed and manufactured for clinical use. The Group has increased its resources and expanded its capabilities to progress these projects underway at VALLAURIX.
Ultimately, the long-term financial objective of the Group is to achieve and maintain sustainable profitability. Key to longer-term profitability is not only continuing the successful research and development of its portfolio of assets but also their successful commercialisation, manufacturing and distribution, and the ability to attract additional funding to support these activities should the need arise.
Environmental Regulations And Performance
The Group’s operations are not regulated by any significant environmental regulation under a law of the Commonwealth, or of a State or Territory, or of any other jurisdiction.
Rounding Of Amounts
The Group is a type of company referred to in ASIC Corporations (Rounding in Financial/Directors’ Reports) Instrument 2016/91 and therefore the amounts contained in this report and in the financial report may have been rounded to the nearest $1,000,000 or in most other cases, to the nearest dollar.
Indemnification And Insurance Of Directors And Officers
During or since the end of the financial year the Group has given or agreed to indemnify, or paid or agreed to pay, insurance premiums to insure each of the Directors against liabilities for costs and expenses incurred by them in defending any legal proceedings arising from their conduct while acting in the capacity of Director of the Group, other than conduct involving wilful breach of duty in relation to the Group. Details of the amount of the premium paid in respect of insurance policies are not disclosed as such disclosure is prohibited under the terms of the contract.
Directors' Benefits And Interest In Contracts
Since the end of the previous financial year no Director has received or become entitled to receive a benefit (other than a benefit included in the total amount of emoluments received or due and receivable by Directors shown in the financial statements and the remuneration report), because of a contract that the Director or a firm of which the Director is a member, or an entity in which the Director has a substantial interest has made with a controlled entity.
Further information on these contracts is included in Note 20 to the financial statements.
REMUNERATION REPORT
REMUNERATION REPORT
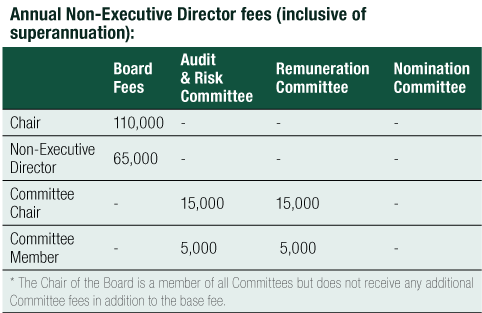
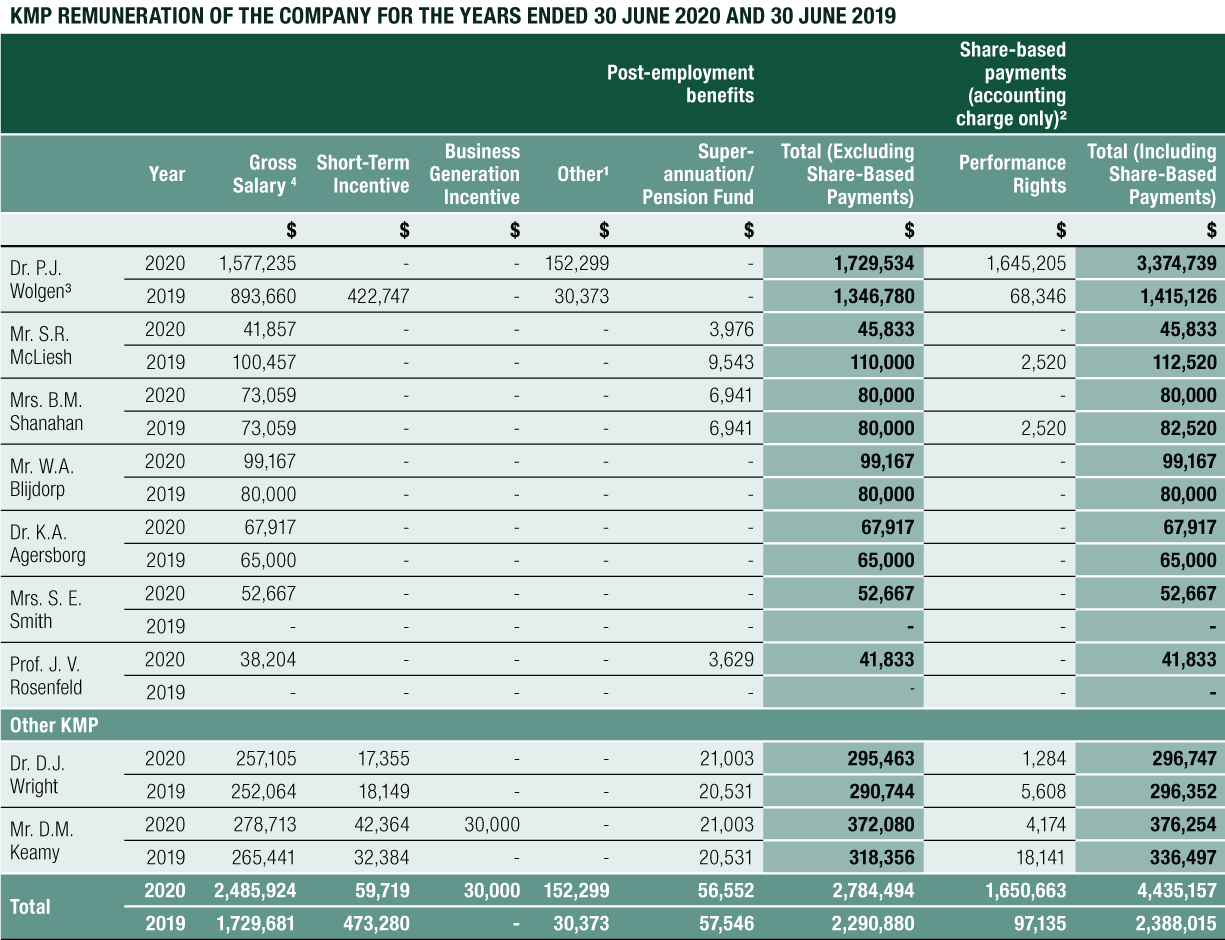
The Remuneration Report, which forms part of the Directors’ Report, provides information about the remuneration of the Directors of CLINUVEL PHARMACEUTICALS LTD and Other Key Management Personnel for the year ended 30 June 2020.
Key Management Personnel (‘KMP’) has the meaning given in the Australian Corporations Act and who together have the authority and responsibility for planning, directing and controlling the activities of the Group, being (see right).
The Remuneration Report is set out under the following main headings:
A. Introduction by the Chair of the Remuneration Committee
B. Remuneration Governance
C. Executive Remuneration
D. Non-Executive Remuneration
E. Service Agreements 2019/20
F. Share Based Remuneration
G. Details of Remuneration
H. Additional Information – Remuneration
A. INTRODUCTION BY THE CHAIR OF THE REMUNERATION COMMITTEE

Chairman of the Remuneration Committee: Mr Willem Blijdorp
As the Chair of the Remuneration Committee, I am pleased to present our Board’ Remuneration Report for the year ended 30 June 2020.
We have kept the format of this year’s Remuneration Report consistent with last year’s since there have not been any significant changes in internal policies or regulations.
I oversee the Group’s remuneration philosophy which aims to attract, retain and motivate talented professionals we require for the Company to meet its strategic objectives. In this process, we wish to see professionals receive appropriate acknowledgement and remuneration.
The Remuneration Committee oversees a remuneration policy of the Group to ensure it is fair, competitive with international peers and transparent. All in all, we strongly believe that a remuneration policy well implemented should help us drive growth strategy and sustainability of CLINUVEL, and thus far we have been proven right.
In 2020, CLINUVEL delivered another year of growth despite the global pandemic and lockdowns affecting all EU countries and the US. Whilst many peer companies have been posting losses and have been required to raise capital, CLINUVEL’s Board and shareholders were fortunate to have a prudent executive management team who had foreseen and planned for the economic downturn and maneuvered the Company through tough times.
In the past year, we increased our revenues by 4%, our cash reserves by 23% and expanded the distribution of SCENESSE® across Europe. Standing tall and with some Dutch pride I look at how CLINUVEL posted an NPAT of A$16.6M and PBIT of A$13.1M in the middle of a global crisis and when many hospitals in Europe and US were not seeing out-patients in the first half of 2020. Our management skilfully came up with a plan to guarantee continuity of treatment for these patients able to travel under the national restrictions imposed by governments.
The Remuneration Committee with consensus of the full Board designed a Performance Rights Plan, which had received shareholder approval in November 2019 and which outlines step by step the Performance Conditions which need to be met for the Company to grow on all fronts within a vesting period of 4 years. This progressive but realistic business plan is being implemented and incentivizes executives and staff along the way. Assuming these Performance Conditions are met the Company should have grown significantly in value, providing commensurate returns for shareholders.
As I had stated in relation to the Committee’s intentions in 2019, this year we reformed the Executive Agreements by eliminating cash-based Business Generating Incentives, and substituting these by a Performance Rights Plan vesting over 4 years. This PRP aims to incentivize and align the interests of the management team with those of shareholders, and in a dual position as Chairman of the Remuneration Committee and also as larger investor of the Company I fully support this direction.
In building this Company, a careful financial and operational approach is taken which is receiving much recognition from the financial community I am in contact with and despite the shortselling we have seen in CLINUVEL the past year. The cost of issuing shares repetitively, the dilution caused to shareholders and the distraction to a management has been successfully avoided, and in terms of risk management we could not have asked for more.
In 2020, we also implemented the decision which forms part of the Company’s policy that non executive directors will not participate in a Performance Rights Plan or Options Scheme since we wish to retain the independence of the non-executive directors.
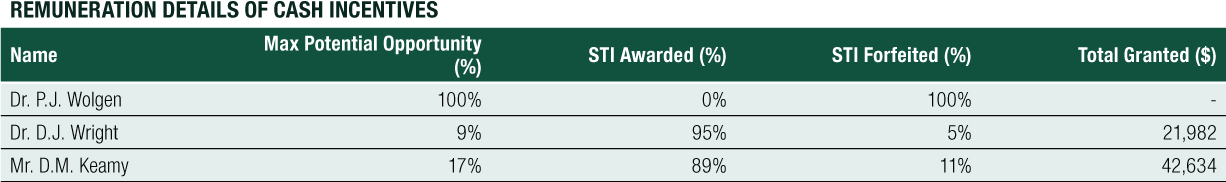
In recent weeks, the Remuneration Committee and Board has been surprised, impressed but proud to learn that our CEO has waived and rejected his STI awarded to him for achieving 70% of the KPIs for 2020. His decision reflects an unusual and amazing leadership in the industry, awareness of the world and extraordinary sensitivities to ask the Remuneration Committee to reinvest these monies for further growth of the Company. In my long career it is rare to find executives who independently pass on their incentives and financial awards for the greater benefit of the Company, shareholders and patients: in Philippe we have long identified a professional of different calibre.
Finally, to summarize in a more personal way, I see the current performance of the Company as follows. The approach and policies of the Remuneration Committee need to change with the times we are living in, and therefore a company and management team needs to be innovative and behave in an entrepreneurial way. For this to succeed, the Company needs to provide a working environment for them to stay on and incentivize them for their achievements.
In CLINUVEL, we have a CFO, CSO and CEO who have this mindset and are not getting out of the boat half-way down the trip: their trip is far from easy, but they have delivered and continue to perform and have built a Company to survive all challenges. As Chairman of the Remuneration Committee this is amazing, I am not sure I would have the stamina to stay on this project for 2 decades.
My vision is simple, entrepreneurship is to implement new combinations, create new business models and change when the environment asks you to do this. With this mindset, this management team is beating all challenges even in the face of economic hard times.
I recommend the Remuneration Report 2020 to all our shareholders and proxy representatives.
Willem Blijdorp, Chairman of the Remuneration Committee
Amsterdam
B. REMUNERATION GOVERNANCE
(i) Remuneration Committee
The Board has provided a mandate to the Remuneration Committee to assist and advise on determining appropriate remuneration policies for its KMP over time, taking into account the relationship between pay and performance, and the results of any evaluations or review processes. The Board has also provided a mandate to the Remuneration Committee to provide advice on non-executive director fees and advice on setting salaries and fees, short- and long-term incentives and employment terms and conditions for its Key executives.
The objectives of the Remunerations Committee’s responsibilities are to ensure that:
a) Remuneration of the Company’s KMP is aligned with the interests of the Company and its shareholders within an appropriate control framework, taking into account the Company’s strategies and risks.
b) The level and composition of remuneration attracts, retains and motivates people of high calibre and with unique specialist industry knowledge to work towards the long-term growth and success of the Company.
c) The role that total fixed remuneration and short- and long-term incentives play is clearly defined and provides a clear relationship between performance and remuneration.
d) The levels and structure of remuneration are benchmarked against relevant peers and considered against global employment market conditions.
e) The Company gives due consideration to applicable legal requirements and appropriate standards of governance.
The methods used by the Remuneration Committee to assess Board performance is disclosed in the Corporate Governance Protocol.
(ii) Remuneration Recommendations
Under the provisions of the Committee’s Charter, the Committee may engage the assistance and advice from external remuneration advisors. To ensure that any recommendations made by remuneration consultants are provided without undue influence being exerted by Executives, external remuneration consultants deliver their advice directly to members of the Committee.
(iii) Voting and feedback at the Company’s last Annual General Meeting
In the 2019 Annual General Meeting (AGM), the Company obtained 88.96% of the proxy votes (including votes at the Board’s discretion) in favour of adopting the 2018/19 Remuneration Report, and this resolution was carried in favour by poll with 87.69% of votes cast. The Company did not receive any further specific feedback at the AGM on its remuneration practices.
(iv) Historical voting at the Company’s Annual General Meetings since 2006
Since 2006 the Company has obtained a historical average of 92%of proxy votes received (including votes at the proxy’s discretion), either carried by a show of hands prior to and including the 2014 AGM or by a poll result after the 2014 AGM, in favour of adopting the Remuneration Reports presented.
C. EXECUTIVE REMUNERATION
(i) Executive Remuneration Framework
The Company’s reward framework has historically provided for a mix of fixed pay and variable pay. The variable pay is structured to incentivise:
- Short-term (generally cash payments in the form of performance-based incentives awarded at a fixed amount or as a percentage of base salary).
- Long-term (generally based upon the issue of performance rights to acquire shares in the Company, and in relation to the Managing Director and to the Chief Financial Officer, other fixed amount cash incentives, including retention awards to recognise ongoing commitment to the Company).
The following diagram links each of the executive remuneration components to the Company’s mission and strategy.
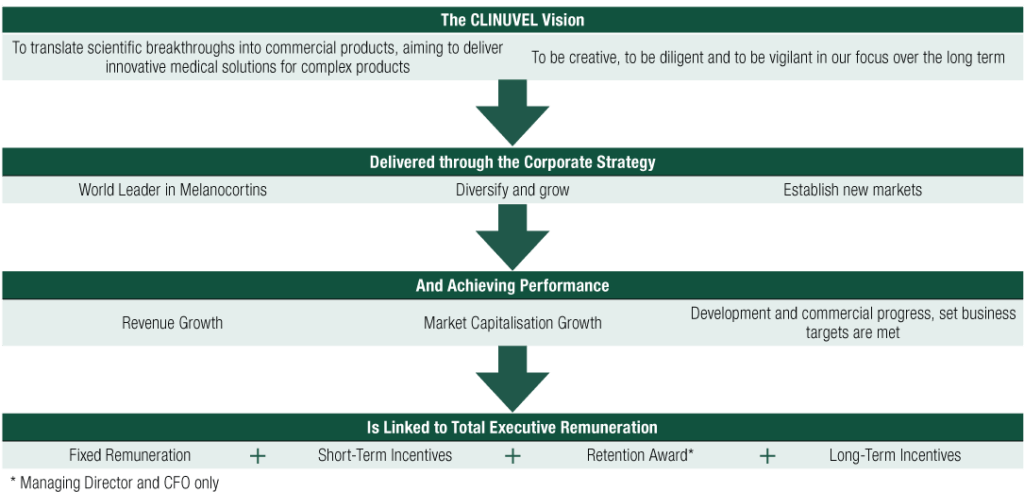
* Managing Director and CFO only
(iii) Managing Director Remuneration – Further Information
The inherent risk of failure within pharmaceutical development is high and this risk is magnified for the Company due to its specialised and narrow focus on developing and commercialising a novel, first-in-class and first-in-line therapies in diseases where there is an unmet clinical need.
The current progress and success of the Company needs to be set against the previous managerial attempts which had posed operational, regulatory and financial challenges. To mitigate the risk and to provide a strong platform to achieve meaningful progress, the Board has followed a business model where most operational skills are retained in-house, where possible, and most management responsibilities are concentrated between the Managing Director (acting in a dual capacity as Chief Executive Officer and Chief Medical Officer) and the Chief Scientific Officer. The Managing Director has the responsibility of guiding and overseeing the execution of the overall corporate strategy, has global responsibility for the safety aspects of the drug (including pharmacovigilance and quality management) and is responsible for commercial drug pricing and reimbursement negotiations. The Chief Scientific Officer is responsible for pre-clinical programs, toxicology, the manufacturing of the drug delivery program, clinical program and setting the regulatory strategies in close coordination with the Board of Directors. As the business evolves and progresses through its development path, this centralised management model will continue to evolve, and key management responsibilities will be shared across new and existing senior management throughout the Group.
The Managing Director’s remuneration structure is reviewed every three years to ensure:
- A maximum level of incentivisation to lead and advance the Company’s program from its current stages of development and commercial growth to serve the long term interest of the Company, taking into account the unique risk and complexity within the business model; and
- It is competitive in international markets, industry and related fields of expertise and providing for specific skillsets.
- longevity of his 15 years of service as CEO compared against local and international peers;
- track record, integrity and professional qualifications for the position;
- the enterprise value created since first employment;
- the shareholder value created in the past three years leading up to the renewal to the service agreement (from 1 July 2016 to 30 June 2019);
- capability to sustain the Company’s focus to maximise profitability following market access; and
- a demonstrated result to attain stability of the business and management team over the long-term.
(iV) Executive Remuneration Benchmarking
One of the objectives of the Remuneration Committee’s responsibilities is to ensure that the levels and structure of remuneration are benchmarked against relevant peers and considered against global employment market conditions. CLINUVEL refers to a select group of publicly listed companies on the ASX and on international securities exchanges for the purpose of peer group analyses. The selection criteria for these companies is broadly based on comparison of:
a) businesses of similar complexity and innovative nature,
b) businesses of similar scope and scale,
c) sectors requiring highly technical and specialised skills,
d) businesses of similar value, reflected in market capitalisation,
e) businesses who have demonstrated similar progress in achieving business outcomes, and
f) businesses of similar risk profile.
CLINUVEL aims to provide competitive remuneration for the Managing Director based on both local and international comparable positions in the relevant market(s). CLINUVEL is a company operating globally with the bulk of its operations and financial exposure falling outside Australia. Its remuneration structure requires to be competitive to international benchmarks.
During the year the Managing Director’s remuneration was benchmarked against 6 revenue-generating Australian-listed life science companies with market capitalisation between $400 million and $2 billion, along with 11 profit-generating US-listed pharmaceutical companies with market capitalisation between US$200 million and US$10 billion. The current total remuneration level of the Managing Director remains below the median level.
(V) Relationship Between Remuneration And Performance
The Group has been solely dedicated to the research, development and commercialisation of its unique and medically beneficial technology. The remuneration and incentive framework, which has been put in place by the Board, has ensured executive personnel are focussed on both maximising short-term operating performance and long-term strategic growth to promote shareholder value. The focus on growth in shareholder value has been centred on achievement of regulatory, development, commercial and operational outcomes, where financial metrics are not necessarily an appropriate measure of executive performance and is commonly expected in other market segments. In recent years the Board has recognised that both financial and non-financial performance measures have been a key link to driving share price performance and this has been reflected in various performance conditions attached to the long-term equity incentives.
The table below shows the progress made in moving through the clinical pathway and into the commercialisation pathway, reflecting the performance of executive management under the leadership of the Managing Director. The table also links to share price performance.
(vi) Executive Remuneration Pay Mix
The Board believes the remuneration mix aligns the Managing Director and Other Executive KMP to shareholder interest. The remuneration mix for 2019/20 is demonstrated as follows:
A comparison of the 3-Year and 5-Year Total Shareholder Return (TSR) of life science peer companies (being a mix of medical device, pharmaceutical product and diagnostic focussed companies) referred above in section (iv) with CLINUVEL’s TSR for the same period shows CLINUVEL is ranked fourth and third respectively.
D. NON-EXECUTIVE REMUNERATION
The Board seeks an appropriate mix of skill, diversity, experience and specific expertise to steward the Company’s success. The Remuneration Committee recommends to the Board individual Non-Executive Director fee levels to attract and retain those with the aforementioned attributes, having regard to global employment market conditions and consultation with specialist remuneration consultants with experience in the healthcare and biotechnology industries.
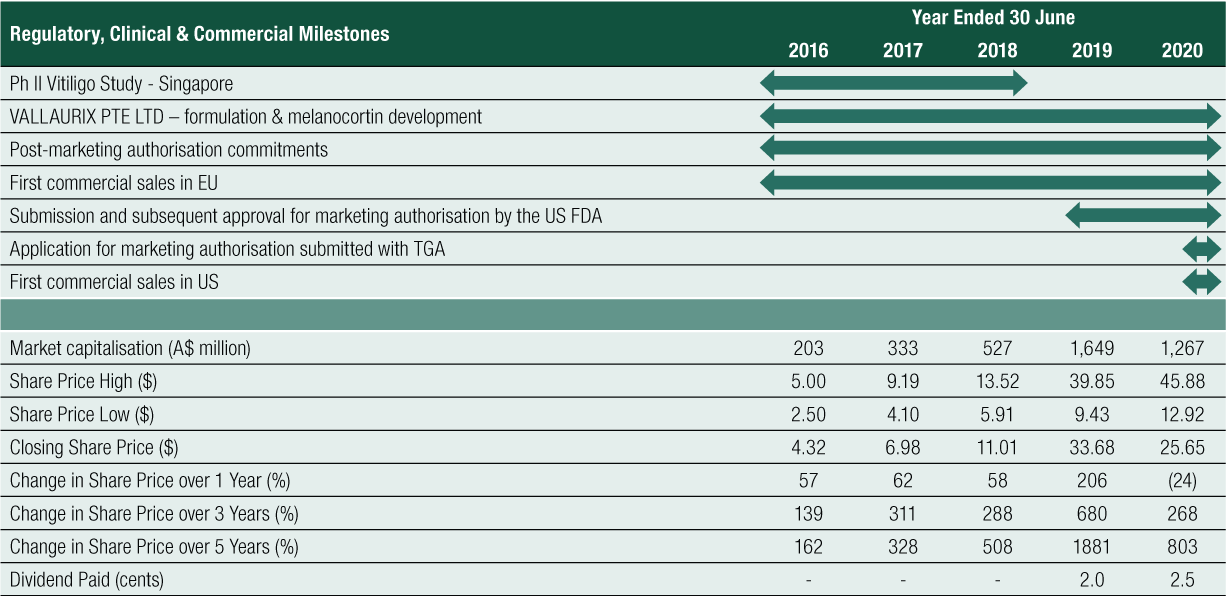
Non-Executive Director Fees
Non-Executive Director fees consist of base fees and committee fees and are inclusive of superannuation and all other contributions. There are no further retirement benefits. The fees are outlined in the table below (see right).
Under the Company’s Constitution, the maximum aggregate remuneration available for division among the Non-Executive Directors is to be determined by the shareholders in a General Meeting and was set at $700,000 at the 2019 AGM. This amount (or some part of it) is to be divided among the Non-Executive Directors as determined by the Board. The aggregate amount paid to Non-Executive Directors for the year ended 30 June 2020 was $387,417.
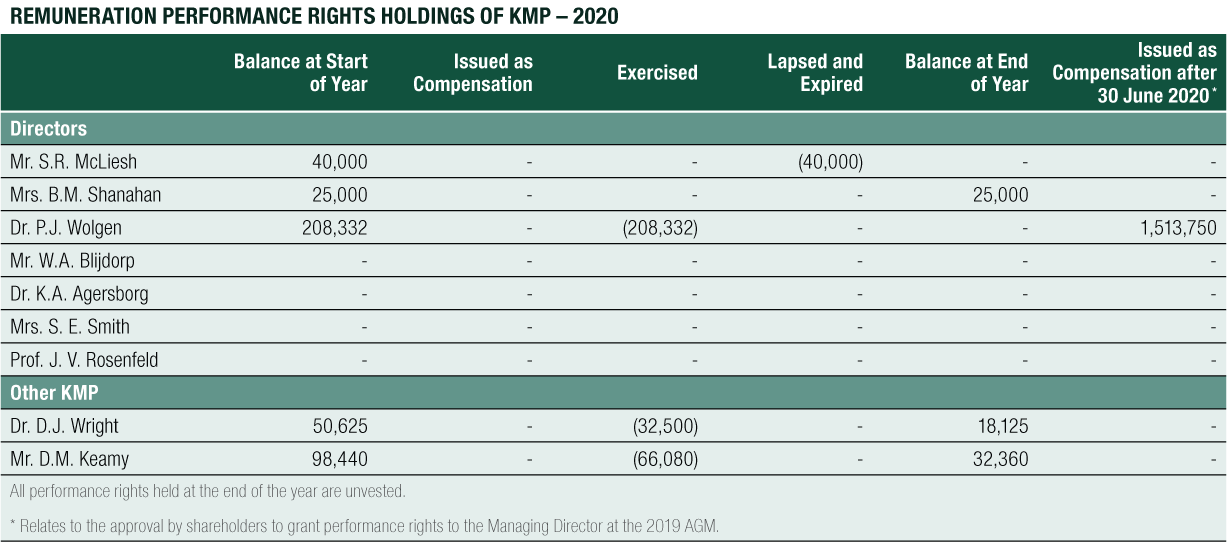
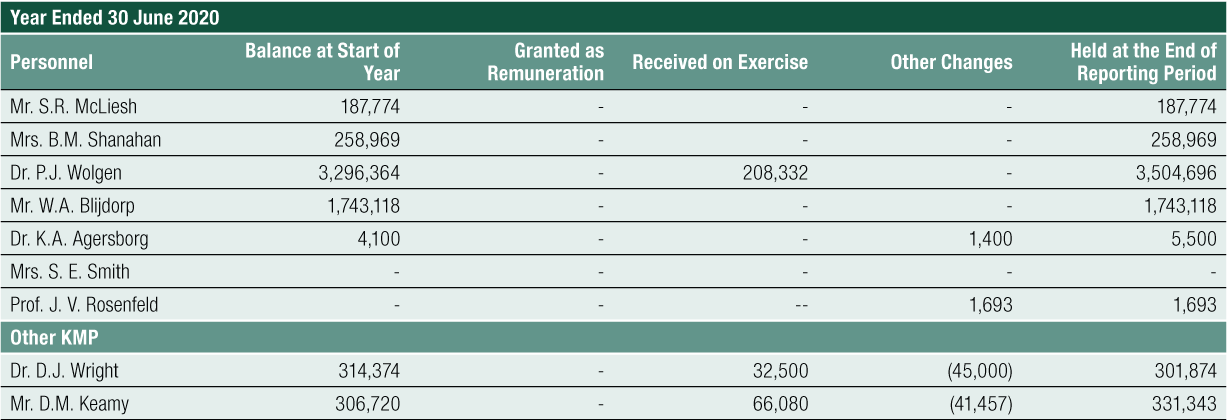
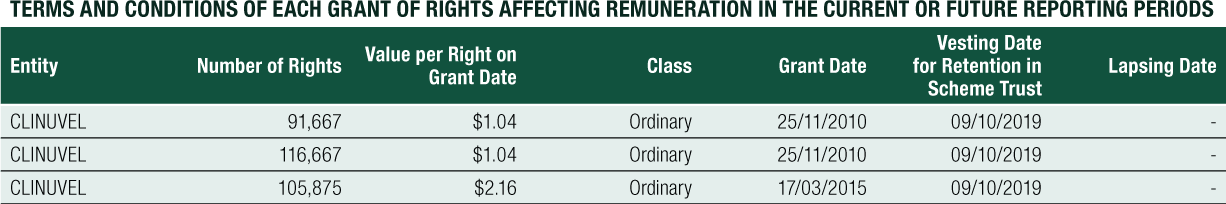
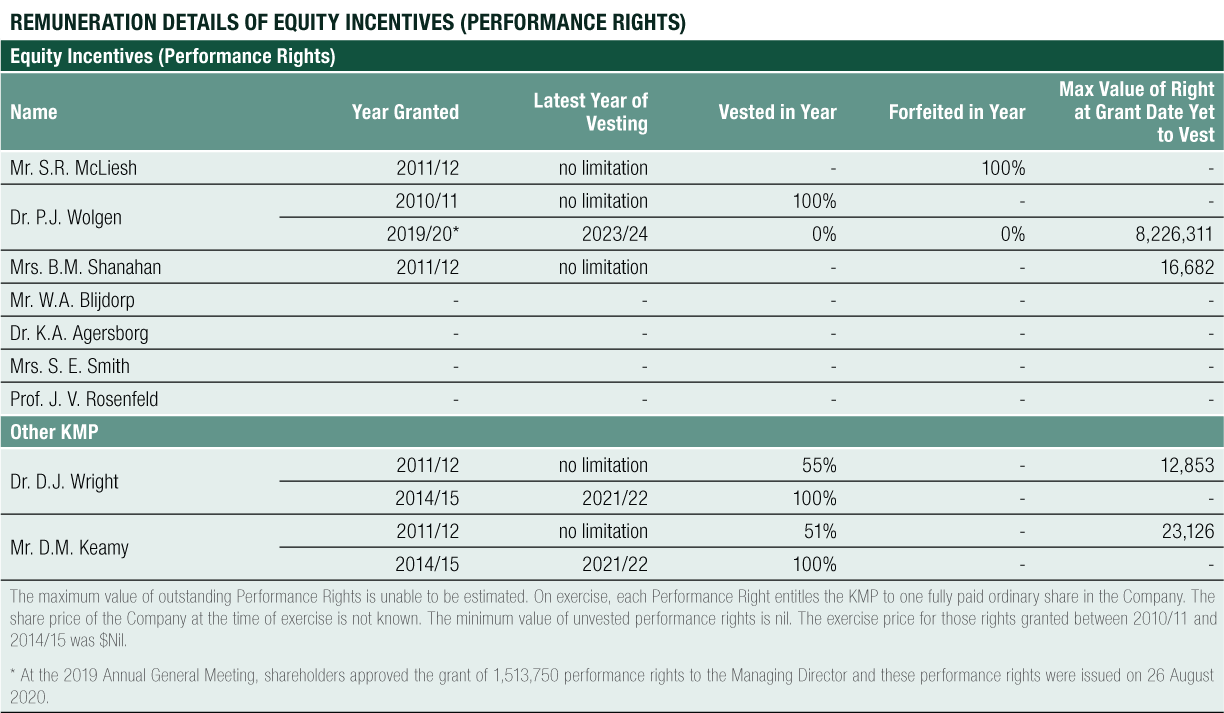
Non-Executive Director Long-Term Incentive – Equity Compensation
The long-term equity remuneration was formerly provided to Non-Executive Directors via the CLINUVEL Conditional Rights Plan and the Performance Rights Plan. Any issue of performance rights to Non-Executive Directors requires shareholder approval.
The Board had previously considered the relatively small management team comparative to peer companies when setting Non-Executive Director remuneration policy. The Board considered that from time to time its Non-Executive Directors would become involved in steering management and engage in certain operational matters that would not commonly be expected of those in a non-executive capacity. Furthermore, the Company ensured the interests of all its KMP, including those in a non-executive capacity, were aligned with the interests of the Company and its shareholders within an appropriate control framework, addressing the preference of some shareholders to see Non-Executive Directors have shareholdings in the Group.
It is no longer planned for Non-Executive Directors to participate in long-term equity compensation plans. Only one current Non-Executive Director in Mrs Shanahan still holds Performance Rights, the last date of issue being November 2014.
E. SERVICE AGREEMENTS 2019/20
Remuneration and other terms of employment for the Managing Director is formalised by a service agreement determined by the Remuneration Committee. The agreement provides for base salary, short- and long-term incentives, other benefits and participation, when eligible, in the CLINUVEL Performance Rights Plan.
The Managing Director, in consultation with the Remuneration Committee, oversees the service agreements entered into with other Executive KMP, providing for base salary, incentives, other benefits and participation, when eligible, in the CLINUVEL Conditional Rights Plan.
On appointment to the Board, all Non-Executive Directors enter into a service agreement with the Company in the form of a letter of appointment. The letter summarises the Board’s policies, the Director’s responsibilities and compensation for holding office. The details of the service agreements to the Managing Director and Executive KMP are:
F. SHARE-BASED REMUNERATION
The Group has an ownership based scheme for Directors4, Other Executive KMP, employees and select consultants of the Company which is designed to provide long-term incentives to deliver long-term value.
Performance Rights
All Performance Rights that have been issued fall under two Performance Rights plans:
a) the CLINUVEL Conditional Performance Rights Scheme (2009); and
b) the CLINUVEL Performance Rights Plan (2014).
152,710 Performance Rights issued under the 2009 Scheme remain unvested as at 30 June 2020 and there are no further performance rights issued under the 2014 Plan remaining unvested at 30 June 2020. 1,513,750 Performance rights were approved by shareholders to grant to the Managing Director at the 2019 AGM under the 2014 Plan. It is no longer intended to issue performance rights under the 2009 Plan.
a) Conditional Performance Rights Scheme (2009)
The Conditional Performance Rights Scheme (2009) is available to eligible employees of the Company. Any issue of rights to Directors requires shareholder approval in accordance with ASX Listing Rules. All rights convert to one ordinary share of the Group and are issued for nil consideration, have no voting rights, are non-transferable and are not listed on the ASX. These can be converted to ordinary shares at any time once the vesting conditions attached to the rights have been achieved, whereby these will be held in a Scheme Trust on behalf of the eligible employee for up to seven years.
The eligible employee can request for shares to be transferred from the Scheme Trust after seven years or at an earlier date if the eligible employee is no longer employed by the Company or all transfer restrictions are satisfied or waived by the Board in its discretion.
4 It is no longer planned for Non-Executive Directors to participate in long-term equity compensation plans. Please refer to ‘Non-Executive Director Long-Term Incentive – Equity Compensation’ in section D to this Remuneration Report
b) Performance Rights Plan (2014)
The Performance Rights Plan (2014) is available to eligible persons of the Company. Any issue of rights to Directors requires shareholder approval in accordance with ASX Listing Rules. All rights convert to one ordinary share of the Group and are issued for nil consideration, have no voting rights, are not listed on the ASX and are non-tradeable (other than with prior written Board consent). They can be converted to ordinary shares at any time once the vesting conditions attached to the rights have been achieved, whereby, at the discretion of the Board, they will be held in a Plan Trust on behalf of the eligible person.
The eligible person cannot trade the shares held by the Plan Trustee without prior written Board consent until the earlier of seven years from grant date of performance rights, when the eligible person ceases employment or when all transfer restrictions are satisfied or waived by the Board in its discretion. Unless the Performance Rights are granted with a shorter vesting period, Performance Rights under this plan lapses after seven years from grant date.
Performance rights are valued for financial reporting purposes using either a Monte Carlo simulation pricing model or a binomial valuation pricing model and are represented as accounting values only in the financial statements. Holders of Performance Rights may or may not receive a benefit from these amounts, either in the current or future reporting periods. The value of all Performance Rights granted, exercised and lapsed during the financial year is detailed in the tables within the Remuneration Report.
H. ADDITIONAL INFORMATION – REMUNERATION
For each cash incentive and right granted, the percentage of the available grant or cash incentive that was paid or vested in the financial year, and the percentage forfeited due to unmet milestones (including service length), is set out below. Cash incentives are paid in the year following the period of performance.
REMUNERATION DETAILS OF EQUITY INCENTIVES (PERFORMANCE RIGHTS)
REMUNERATION DETAILS OF CASH INCENTIVES
LOANS TO DIRECTORS AND EXECUTIVES
No loans were granted to Directors or executives for the years ended 30 June 2020 and 30 June 2019.
SHARES PROVIDED UPON EXERCISE OF RIGHTS
DETAILS OF SHARES ISSUED DURING THE FINANCIAL YEAR AS A RESULT OF EXERCISE OF RIGHTS
DETAILSOF SHARES ISSUED DURING THE YEAR TO EMPLOYEES FROM THE 2009 SCHEME TRUST AND THE 2014 PLAN TRUST
NON-AUDIT SERVICES
For the year ended 30 June 2020, Grant Thornton Australia provided audit services to the Company. Grant Thornton Australia also provided non-audit services, specifically tax related services. Details of amounts paid or payable to the auditor for non-audit services provided during the year by the auditor are outlined in Note 19 to the financial statements.
The Directors are satisfied that the provision of non-audit services, during the year by the auditor is compatible with the general standard of independence for auditors imposed by the Corporations Act 2001. The Directors are of the opinion that the services as disclosed in Note 19 to the financial statements do not compromise the external auditor’s independence, based on advice received from the Audit Committee, for the following reasons:
- all non-audit services have been reviewed and approved to ensure that they do not impact the integrity and objectivity of the auditor; and
- none of the services undermine the general principles relating to auditor independence as set out in APES 110 ‘Code of Ethics for Professional Accountants’ issued by the Accounting Professional & Ethical Standards Board, including reviewing or auditing the auditor’s own work, acting in a management or decision-making capacity for the Company, acting as advocate for the Company or jointly sharing economic risks and rewards.
AUDITOR’S INDEPENDENCE DECLARATION
The auditor’s independence declaration as required by s.307C of the Corporations Act 2001 is included and forms part of this Directors’ Report.
PROCEEDINGS ON BEHALF OF THE COMPANY
No person has applied for leave of Court to bring proceedings on behalf of the Company or intervene in any proceedings to which the Company is party for the purpose of taking responsibility on behalf of the Company for all or any part of those proceedings.
The Company was not party to any such proceedings during the year.
Signed in accordance with a resolution of the Board of Directors pursuant to s.298(2) of The Corporations Act 2001.
Dr. Philippe Wolgen, MBA MD Director
Dated this 26th day of August, 2020
STATEMENT OF PROFIT AND OTHER COMPREHENSIVE INCOME FOR THE YEAR ENDED 30 JUNE 2020
STATEMENT OF FINANCIAL POSITION AS AT 30 JUNE 2020
STATEMENT OF CASH FLOWS FOR THE YEAR ENDED 30 JUNE 2020
STATEMENT OF CHANGES IN EQUITY FOR THE YEAR ENDED 30 JUNE 2020
NOTES TO AND FORMING PART OF THE FINANCIAL STATEMENTS FOR THE YEAR ENDED 30 JUNE 2020
NOTES TO AND FORMING PART OF THE FINANCIAL STATEMENTS FOR THE YEAR ENDED 30 JUNE 2020
DIRECTORS’ DECLARATION
DIRECTORS’ DECLARATION
In the opinion of the Directors:
- the financial statements and notes of the consolidated entity are in accordance with the Corporations Act 2001, including:
a) giving a true and fair view of the consolidated entity’s financial position as at 30 June 2020 and of its performance for the year ended on that date; and
b) complying with Accounting Standards; and
c) complying with International Financial Reporting Standards as disclosed in Note 1
- there are reasonable grounds to believe that the Company will be able to pay its debts as and when they become due and payable; and
- the audited remuneration disclosures set out in pages 36 to 47 of the Directors Report comply with Section 300A of the Corporations Act 2001.
This declaration is made in accordance with a resolution of the Board of Directors. The Directors have been given the declarations by the Chief Executive Officer and Chief Financial Officer required by Section 295A of the Corporations Act 2001.
Dr. Philippe Wolgen, MBA MD Director
Dated this 26th day of August, 2020
Shareholder Information As At 18 September 2020
Shareholder Information As At 18 September 2020
Additional information as at 18 September 2020 required by the ASX and not shown elsewhere in this report is as follows:
1. SHAREHOLDING
A) Distribution of shareholder numbers
Ordinary fully paid shares
B) shareholdings held in less than marketable parcels
C) Substantial shareholdings
D) Voting rights
E) Largest shareholders
2. COMPANY SECRETARY
The name of the Company Secretary is:
Darren Keamy
3. REGISTERED OFFICE
The principle registered office in Australia is:
Level 11, 535 Bourke Street
Melbourne, VIC 3000, Australia Telephone: +61 3 9660 4900
Fax: +61 3 9660 4999
Email: [email protected]
Website: http://www.clinuvel.com
4. REGISTER OF SECURITIES
Computershare Investor Services Pty Ltd
Yarra Falls, 453 Johnston St, Abbotsford,
VIC 3067, Australia
Telephone: +61 3 9415 4000
5. AUSTRALIAN SECURITIES EXCHANGE LIMITED
Quotation has been granted for all the ordinary shares on all Member Exchanges of the Australian Securities Exchange Limited (ASX):
The Company’s shares are also traded on XETRA, an electronic trading system, based in Frankfurt, Germany, under the code UR9.
In the USA, the Company’s Level 1, American Depositary Receipts (ADRs), trade under the code CLVLY. Each ADR of the Company is equivalent to one ordinary share of the Company, as traded on the ASX. The Bank of New York Mellon is the depositary bank.
6. RESTRICTED SECURITIES
Restricted securities on issue at June 30, 2020: Nil.
7. DIRECTORY
NON-EXECUTIVE CHAIR
Willem Blijdorp
NON-EXECUTIVE DIRECTORS
Brenda Shanahan, Dr Karen Agersborg, Susan Smith, Prof Jeffrey Rosenfeld
MANAGING DIRECTOR AND CHIEF EXECUTIVE OFFICER
Dr Philippe Wolgen
CHIEF SCIENTIFIC OFFICER
Dr Dennis Wright
CHIEF FINANCIAL OFFICER AND COMPANY SECRETARY
Darren Keamy
AUDITOR
Grant Thornton Australia Limited
Collins Square, Tower 5, Level 22, 727 Collins Street, Melbourne, VIC 3008, Australia
BANKER
National Australia Bank (NAB)
Western Branch, 460 Collins St, Melbourne, VIC 3000, Australia
LEGAL COUNSEL
Arnold Bloch Leibler
Level 21, 333 Collins St, Melbourne, VIC 3000, Australia
Sidley Austin LLP
Woolgate Exchange, 25 Basinghall Street,
London, EC2V 5HA, United Kingdom
IP LAWYER
Dipl.-Ing Peter Farago
Baadestr 3, Munich 80, Germany