Tables
STATEMENT OF PROFIT AND OTHER COMPREHENSIVE INCOME FOR THE YEAR ENDED 30 JUNE 2020
SHARES PROVIDED UPON EXERCISE OF RIGHTS
DETAILS OF SHARES ISSUED DURING THE FINANCIAL YEAR AS A RESULT OF EXERCISE OF RIGHTS
DETAILSOF SHARES ISSUED DURING THE YEAR TO EMPLOYEES FROM THE 2009 SCHEME TRUST AND THE 2014 PLAN TRUST
REMUNERATION DETAILS OF CASH INCENTIVES
ADDITIONAL INFORMATION – REMUNERATION
REMUNERATION DETAILS OF EQUITY INCENTIVES (PERFORMANCE RIGHTS)
SHARES HELD BY KMP
TERMS AND CONDITIONS OF EACH GRANT OF RIGHTS AFFECTING REMUNERATION IN THE CURRENT OR FUTURE REPORTING PERIODS
DETAILS OF REMUNERATION
KMP REMUNERATION OF THE COMPANY FOR THE YEARS ENDED 30 JUNE 2020 AND 30 JUNE 2019
REMUNERATION PERFORMANCE RIGHTS HOLDINGS OF KMP - 2020
E. SERVICE AGREEMENTS 2019/20
Top Banner
Annual Report
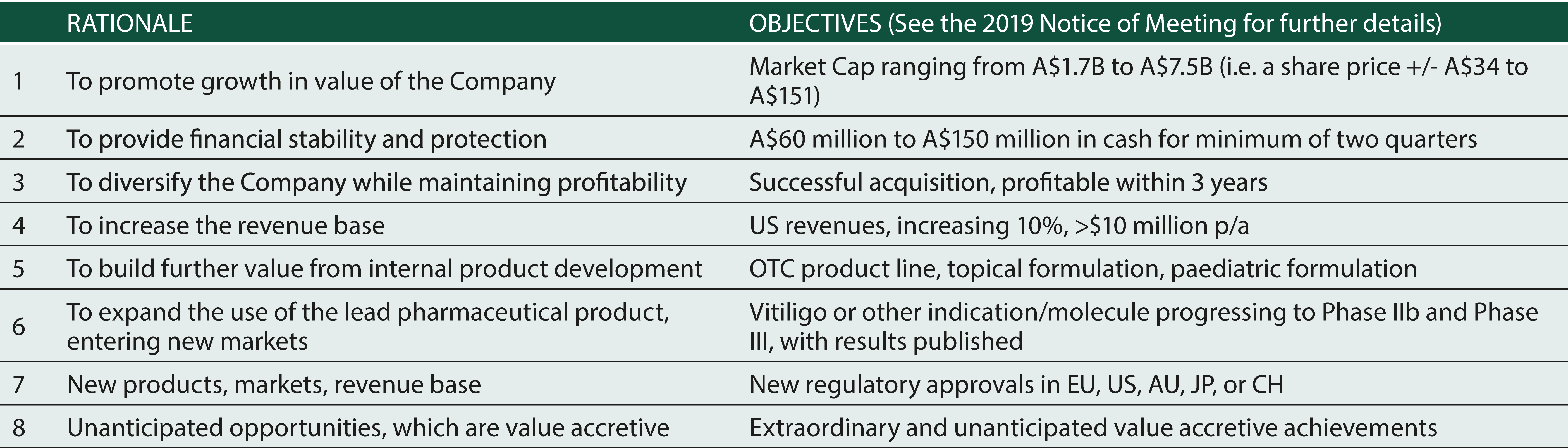
GROWTH AND EXPANSION
GROWTH AND EXPANSION
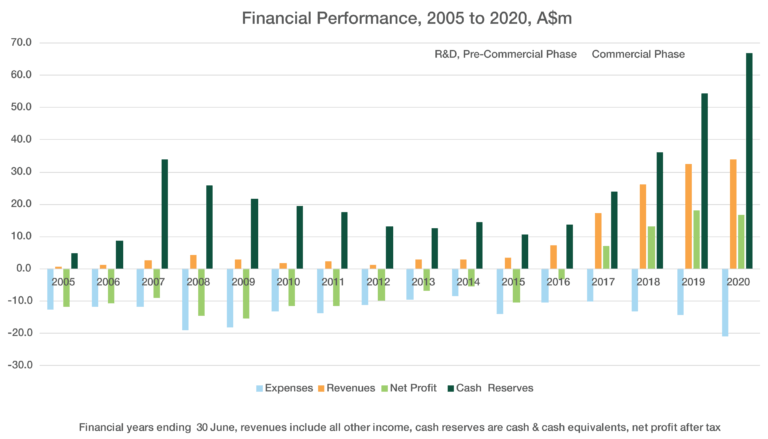
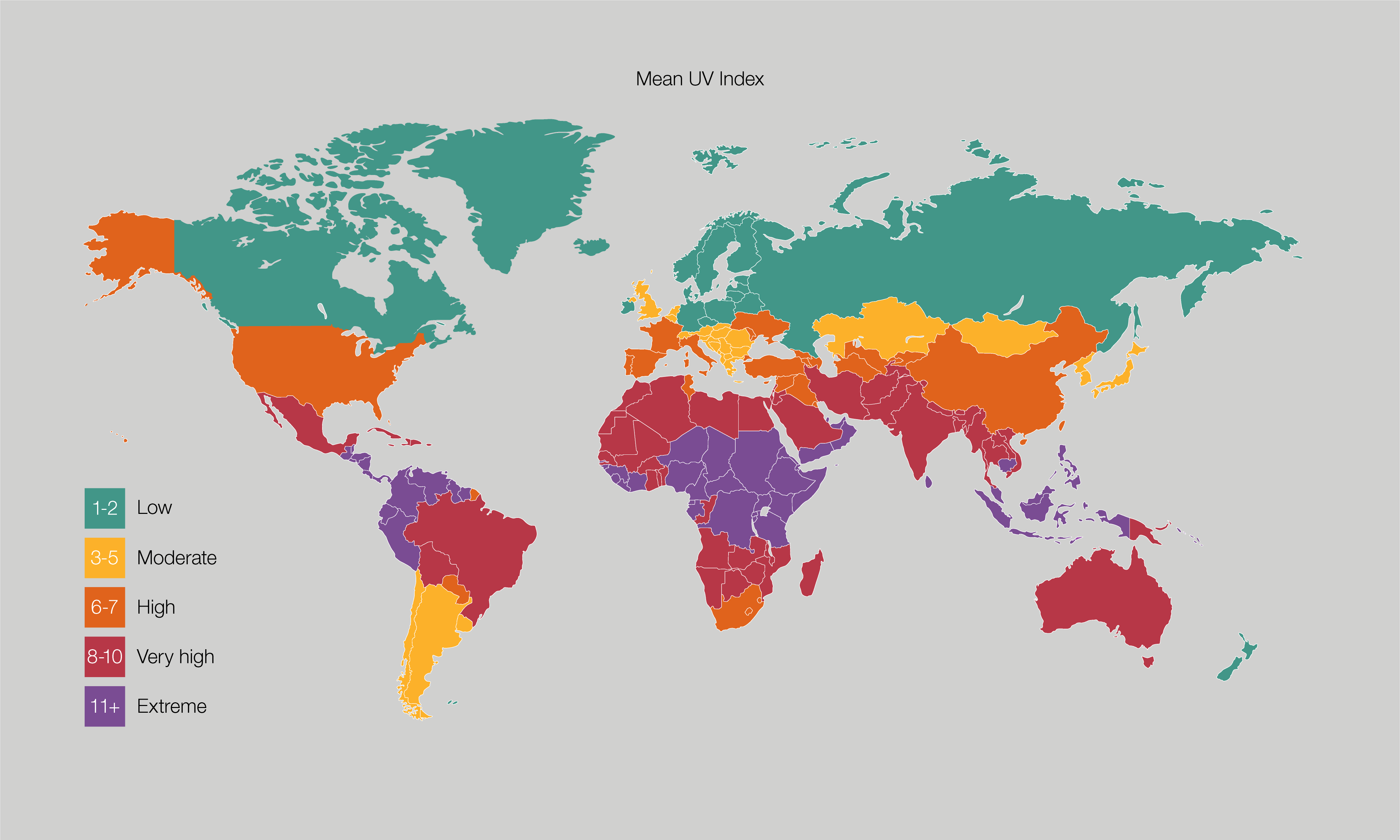
FIFTEEN YEAR PERFORMANCE OVERVIEW
FIFTEEN YEAR PERFORMANCE OVERVIEW
CLINUVEL completed its fourth full year of commercial operations in the financial year ending 30 June 2020, recording a fourth consecutive annual profit and declaring a third consecutive annual dividend.
A STORY OF STRATEGIC FOCUS
Over the last fifteen years, CLINUVEL has implemented a deliberate and focussed strategy to develop and commercialise one lead drug embodying a novel technology, for an unmet need. CLINUVEL has delivered on the strategic objective set in 2005 to develop and commercialise SCENESSE® (afamelanotide 16mg) which has been approved and launched for adult patients with erythropoietic protoporphyria (EPP) in Europe and the USA. Over the financial years 2005 to 2016, CLINUVEL’s operations were research and development (R&D) focussed. During this R&D phase, expenses exceeded revenues as SCENESSE® was developed; clinical studies undertaken and completed; and regulatory approvals, sought and obtained. To finance the business during this time, over A$94m was raised from capital markets in several installments. CLINUVEL’s last capital raising was in March 2016.
COMMERCIALISATION HAS DRIVEN REVENUES, PROFITS AND CASH RESERVES
Commercial operations commenced in Europe in June 2016 following the granting of marketing authorisation by the European Commission in 2014. In the first full year of commercial operations, FY2017, the Company’s first net profit after tax of A$7.1 million was recorded. This outcome was achieved at a higher level in FY2018 (A$13.2 million) and in FY2019 (A$18.1 million) which was a record annual profit. Revenues from operations rose by 5% in FY2020 and there was a deliberate and controlled rise of 44% in expenditure
to support the expansion of the Group’s activities, including establishing commercial infrastructure in the USA following the approval of SCENESSE® by the US Food and Drug Administration (FDA). The Group delivered its fourth consecutive annual profit in FY2020, A$16.6 million. This is a positive outcome, achieved as the world has been adversely impacted by the coronavirus pandemic and resulting significant economic contraction in the second half of the year.
The Company has maintained a disciplined and conservative approach to resource management throughout its development cycle and over the last four years of commercial operations, ensuring that it is capable of withstanding global downturns, removing the need to raise capital in adverse markets and allowing the Group to self-finance its planned expansion and growth. The rise in cash reserves to A$66.7 million as of 30 June 2020 represents a 44% annual compound growth rate since the last capital raising in March 2016. This level of cash reserves covers more than three years of the Group’s FY2020 expenses.
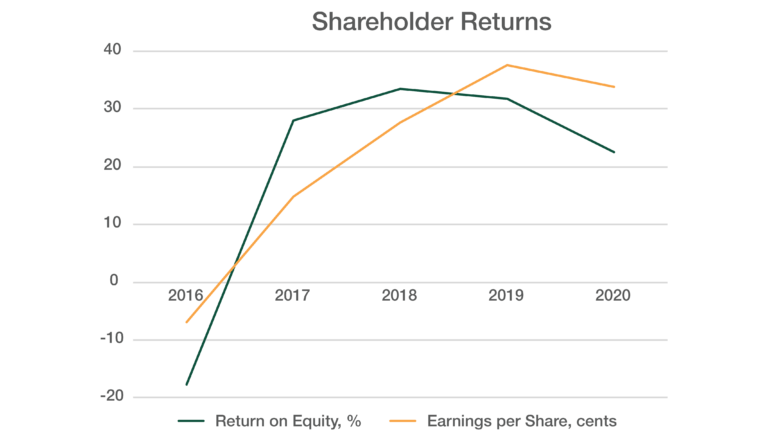
RETURNS TO SHAREHOLDERS
Given the level of cash reserves accumulated, the Board has been able to show its appreciation of the support of shareholders over the R&D phase of operations and into the commercial phase by declaring an unfranked dividend in each of the last three financial years – A$0.02 following FY2018 and A$0.025 after each of FY2019 and FY2020. As a company with positive annual net cash flow and profitability, CLINUVEL has provided shareholders with a positive return on equity and earnings per share over the last four years – 23% and 33.8 Australian cents, respectively in FY2020.
MILESTONE FDA DECISION
FY2020 is notable for the October 2019 milestone decision of the US FDA to approve SCENESSE® for the increase of ‘pain-free’ light exposure in adult EPP patients with a history of phototoxicity. Within six months of this approval, CLINUVEL commenced first treatment of US patients in April 2020 enabling first US revenues in FY2020. CLINUVEL is committed to providing EPP patients worldwide with access to treatment and is actively pursuing regulatory approvals in other jurisdictions.
Approvals from the world’s leading regulatory bodies in the USA and Europe – validating the positive safety profile of SCENESSE® – enable the expansion and increase in pace of the Group’s product development pipeline. CLINUVEL has identified other indications where afamelanotide may provide clinical benefit to patients. This is central to the evolution of the Group’s strategy to become a diversified biopharmaceutical providing treatments to multiple patient groups. The feature in this report on the Expansion of Technology explains CLINUVEL’s path to this objective
SUMMARY
CLINUVEL is a dynamic biopharmaceutical group of companies that has developed and commercialised a novel treatment from laboratory bench to patient treatment, with a track record of positive cash flow and profit since the commencement of commercial operations. This is an achievement of which stakeholders can be proud. This Annual Report provides more detail on the result for FY2020, how it was achieved and the strategies of the Group to grow and expand its activities.
CLINUVEL’S MISSION
The CLINUVEL Group works to translate scientific concepts and breakthroughs into commercial products. We are determined in our desire to excel scientific research and development, building on our global expertise to deliver lifelong care and novel products for patients and consumers. The CLINUVEL Group places much emphasis on its People and Environment as central to all of the Group’s working practise. CLINUVEL Group focuses its research and development on genetic metabolic and diseases not yet addressed, aiming to deliver innovative medical solutions for complex problems.
CLINUVEL’S VISION
Innovating novel solutions for unmet patient and healthcare needs.
CLINUVEL’S VALUES
PEOPLE & ENVIRONMENT
We work for physicians, consumers and our stakeholders. We are selective and invest time in the talent we employ. We aspire to create an environment where professionals are able to develop and grow. We aim to present skilled talent with early opportunities, responsibilities and accountability as part of training the next generation. We strive to build international teams and operate on the basis of gender and ethnic equality. We wish to set an example of excellence in our industry.
TECHNOLOGY
We create, develop, and advance products which are driven by medical need, consumer demand or lack of available solutions. Our technologies aim to add value beyond existing offerings. We acknowledge that new technologies require regulatory environments to be primed and markets to be prepared for achieving widespread acceptance and adoption.
APPROACH
We aim to be innovative in our approach and find solutions for unique, complex and previously neglected healthcare problems. We are determined to remain leaders in our field of expertise, and be creative and diligent in all our endeavours. We admit errors, recognise our shortfalls, evaluate, analyse and learn to implement new findings. In improving ourselves we strive to enhance the lives and quality of life of those we serve. We are vigilant not to become complacent and recognise that success can only come from the identification and mastering of obstacles. Our staff are optimistic and focused.
RESPECT & APPRECIATION
We are conscious of the privilege to be productive during our professional lives. We appreciate the significance of being able to function in good health and we value this gift every day. We aim to be sincere in our approach and represent data and facts. We act respectfully and do not harm others. We value our colleagues and co-workers and cherish diversity, equality, respect and harmony. We are passionate towards our objectives and share empathy and compassion for all those we work to serve.
KNOWLEDGE BUILDING & SHARING
We are experts in optical physics, the interaction of light and human biology, and proficient in our understanding of rare disorders and skin care. We advance our ideas and concepts and translate them into effective and practical solutions. We aim to grow our knowhow continuously and establish a learned community. Collaboratively we seek to excel in a multifaceted field to arrive at scientific breakthroughs.
CHAIR’S LETTER
CHAIR'S LETTER

Dear Shareholders,
Which expert in business could have predicted the year 2020? The question is, were we caught by surprise and could we have managed ourselves better? The year 2020, my first complete year as Chairman of CLINUVEL, has been overshadowed by the impact of COVID-19, and has therefore seen my main tasks change, helping navigate CLINUVEL during these uncertain times.
The financial year was one of two halves. In the first half, CLINUVEL’s goal was to provide continued growth in Europe and to progress our product development pipeline. We concluded the year on a high, with a unique milestone on 8 October 2019, which was the approval by the US Food and Drug Administration (FDA) to make SCENESSE® (afamelanotide 16mg) available in the USA to adult EPP patients. We shared our celebration at the 2019 Annual General Meeting (AGM).
With much optimism, we looked forward to expanding the business into the USA, progressing our product development pipeline as we started the second half of the 2020 financial year. In January 2020, our IR Manager, Malcolm Bull, came back with considerable optimism from the JP Morgan Global Healthcare Conference in San Francisco. Then suddenly, a dramatic turn of events which turned the world upside down. In this new world, making sure that all checks and balances were crossed, my priorities became threefold: to oversee the governance and leadership, to optimise the financial management, and to make sure unique people with great knowledge would continue with this Company.
Governance and Leadership
I was asked how I identify executive managers to run my portfolio of companies. In my own publicly listed company BSGR, I have recently appointed a new CEO who is stress-resistant and fully understands the art of managing people to maximise their skillsets. Captains of industries are truly identified when they face changing environments and are forced to act. Therefore, this past year was particularly important to closely watch how CLINUVEL’s senior managers would handle the crises and take the most suitable measures. Entrepreneurship is much more than running successful companies when all things are going well. I expect senior management to find creative solutions, come up with ideas on how to progress a company and, above all, dare to take unpopular measures to retain value. In the past year I have again seen proof of CLINUVEL’s leadership, who managed the complex world of pharmaceuticals when maneuvering was really needed. At a time when the world went in lockdown and hospitals decided to put COVID first and other healthcare services second, our team managed to maintain supply and maintain SCENESSE® treatment.
In CLINUVEL, we have passed more than a decade of independent audits without remarkable findings.
With respect to governance, the Board sets clear goals. We have split the role of the CEO and Chairman. We did not just delegate all the responsibilities to one person, helping the Managing Director get on with his daily job of running the Company and leave the supervisory matters to myself, as well as the coordination of Board matters. In many listed companies the roles are still combined, but I think it is not a wise way of running companies.
I see success in business as a simple formula which has always worked for my companies and most successful colleagues. First, always make sure you spend less than you receive. Second, formulate a clear vision and work consistently towards this goal. Third, treat the people working around you with respect and they will do the same through putting in maximum effort. With our current CEO, we see all three.
Effective leadership is not just one person, but it is a culture created where collaboration takes place across all managers on the floor and business partners without issues or friction. In this interplay, the CEO then needs to integrate a mix of long term and new employees, guarantee diversity, possess a depth of skills and experience and report progress to go through all bottlenecks. In the last 12 months, we have expanded our workforce considerably and are all working to achieve a list of our key objectives.
Financial Leadership
What is financial leadership and why does it matter? The worst business one can arrive in is a business where processes are lacking, expenditures are varying, and little financial control exists due to high commitments of forced spending. The financial books of a company are the reflection of managers’ ability to make daily decisions. CLINUVEL’s books are not coincidental, they follow our managers’ vision.
It is clear CLINUVEL’s financial management has been very successful, both our CFO and CEO protect our financial position to a maximum.
I always wish to see clean financial processes in the companies in which I am involved. In CLINUVEL, we have passed more than a decade of independent audits without remarkable findings. This helps us concentrate on our operations. The CFO Darren Keamy has built a strong team over the years and oversees the finances of all the activities in the subsidiaries and tax. His team is managing the expansion of VALLAURIX in Singapore, and the currencies across the Company. As part of the team’s task this year, there was a need to avoid negative interest rates on the Euro and secure our working capital.
It is clear CLINUVEL’s financial management has been very successful, both our CFO and CEO protect our financial position to a maximum. We need lean companies in this industry, and the way our management is managing this globally is the modern way to do it. We shareholders benefit long-term, since no money is leaking, and no new capital needs to be raised from the markets. I have seen many investment banks offering to raise money for CLINUVEL at high discount percentages and praise ourselves that we do not need to do it.
Cash is always king in crises and this year I have seen established companies going out of business. Many tragedies are affecting breadwinners and families. With CLINUVEL, we expand and provide people strong job security, helping our employees feel safe and motivated. The way our financial team is going about the business is the right one and as a major shareholder in the Company, I am grateful that we are rock-steady in management without unexpected or dramatic events.
For the six months to December 31 we recorded a good profit. At the end of our audited financial year, we posted a A$16.6 million profit, whereby our cash reserves have substantially grown. As a Chairman, I cannot ask more from a team during such a global financial crisis and am very happy to be involved in such a Company.
In our senior management team, we have 57% women and 43% men, a great balance based on our CEO’s aim to lead in this field.
CONTINUATION OF SPECIAL KNOWLEDGE
CLINUVEL stands out with a special team of Directors, each of whom brings special expertise, and an exact gender balance. CLINUVEL leads many other companies in this balance, showing our different approach to business. In our senior management team, we have 57% women and 43% men, a great balance based on our CEO’s aim to lead in this field.
I am a general entrepreneur without special knowledge about pharmaceutical development, but the same business principles apply to every one of my ventures. I want to see CLINUVEL keep its key people and educate new talent for the years to come. In CLINUVEL, we have become an institution whereby new graduates have started to apply for jobs since the universities have taken the CLINUVEL example of running a company in their business cases. I want to see long-term continuation in an environment where competitors are trying to emulate our approach. A strong top and talented below within our levels of hierarchy is the only way to build a company for generations to come. Looking back at the progress this year, we are well on course.
In CLINUVEL, we need special knowledge to grow, and in Singapore we are increasing our investments each year. This year we built modern facilities which are featured in subsequent pages of this report. The Singapore government started to recognise our investments in the country and our hiring of people to lead the research of new products.
In March, I shared my vision of the Company’s strategy and in October our CEO gave a detailed breakdown of how CLINUVEL will be growing. Philippe has made it clear that he has very little time to waste to perform with his team, since he wants all objectives of the Group to be met before he retires from this role. I understand his ambition is to see the name CLINUVEL strive during his time with the Company, which translates well for us shareholders. I share his frustration in a COVID-19 world, but I have confidence in him and the management team to keep surprising and impressing us, with their dedication to the Company.
I want to thank the CLINUVEL Board for the great support and guidance this year, especially during such a peculiar year, but nonetheless with great highlights.
During these difficult and uncertain times, we sympathise with all those affected by COVID-19 and hope all our stakeholders remain safe and well.
IN CONCLUSION
During these difficult and uncertain times, we sympathise with all those affected by COVID-19 and hope all our stakeholders remain safe and well. We appreciate the loyalty of our long-term shareholders and the new shareholders who have joined CUV during the past year for the ongoing journey. Despite the current adversity, CLINUVEL’s future is full of optimism and we will continue to focus without relaxing on the strategic path we have laid out. This path will lead us to adding value, pride, and satisfaction to all our stakeholders.
Willem Blijdorp
Chairman
CORPORATE GOVERNANCE
CLINUVEL Pharmaceuticals Ltd and its Board are committed to establishing and achieving the highest standards of corporate governance. The Company’s Corporate Governance Statement for the year ending 30 June 2020, based on the Australian Securities Exchange Corporate Governance Council’s (ASXCGC) Corporate Governance Principles and Recommendations, 3rd Edition, can be found on our website at https://www.clinuvel.com/clinuvel/company-overview/corporate-governance.
CLINUVEL IN THE MEDIA
MANAGING DIRECTOR’S LETTER
MANAGING DIRECTOR'S LETTER

Dear Shareholders,
INTRODUCTION
I contemplate CLINUVEL’s successful progress and financial results in 2020 in contrast with the unforeseen pandemic and various containment measures unfolding around the world. It has been a surrealistic series of events in the past year, the emergence of a new epoch and existence for all of us. In the vortex of a virus working its way methodically across all borders and has already stolen 1,000,000 lives around the world, global commerce has slowed down at a rate far beyond that seen during the Great Recession and has caused millions of workers to be laid off.
Like most businesses, CLINUVEL was forced to adapt quickly and plan its operations in a materially different way. First, Brexit caused us to relocate some of our operations to the European Economic Area while keeping other services in the UK. We made this transition successfully and kept adding new services and structures to EU distribution.
…we were proud to learn in February that CUV’s Board was one of only 11 on the S&P/ASX-200 where at least half of the directors were female, reflecting the progressive and intentional approach to gender and cultural spread within the Company.
The past year taught us that limited travel required an indirect approach to CLINUVEL’s operations, and the curfew and inhibited ability to enter offices forced the Board and management to rethink strategy and the future of the Company. Senior management found itself defending the healthcare needs of the targeted porphyria patients both in Europe and the US. While some erythropoietic protoporphyria (EPP) patients could not gain access to treatment due to closure of specialist hospitals, other centres needed to be immediately activated to prescribe SCENESSE®. Under extraordinary circumstances, special permissions were requested from, and granted by, national competent authorities in the European Union and the US.
Following the FDA’s approval in October 2019, there has not been a second to reflect upon – let alone celebrate – the momentous occasion. Rather, it has been an accelerated path to formulate the second product PRÉNUMBRA®, contest Ethics Committees and regulatory authorities, and introduce SCENESSE® to US insurers, all while expanding the Company on all other fronts. We invested in new facilities in Singapore, and despite a delay of several months, were able to open our laboratories on 31 August. With newly elected Chairman, Mr Willem Blijdorp and the addition of Mrs Sue Smith and Professor Jeffrey Rosenfeld as Non-Executive Directors to the Board, new insights, new strategic discussions, and different practices were introduced. The current Board with Mrs Brenda Shanahan and Dr Karen Agersborg is well diversified in skills, and we were proud to learn in February that CUV’s Board was one of only 11 on the S&P/ASX-200 where at least half of the directors were female, reflecting the progressive and intentional approach to gender and cultural spread within the Company.
In terms of cost-efficiency, we have continued to improve with operating expenditure as a percentage of total revenues reaching 26.9%. We also saw an improvement in our overall return on invested capital to 14.2% this year (up from 13.7% in 2019). This performance demonstrates considerable momentum as we enter 2021. We built and kept our balance sheet clean to create options for the ensuing years, increased our cash reserves by 23% and expanded the Group with an eye to overall sustainability. Along a chartered course, our R&D activities gathered momentum to provide for the next generation of products. I see CLINUVEL’s output the past year as one of the most memorable achievements by our dedicated team in the midst of the most severe economic recession in recent history.
As much as we could, we changed our pharmaceutical product supply and worked for weeks 24/7 to secure continued distribution. We witnessed how authorities, distribution centres and customs at various borders were tapering or shuttering operations all together. As supply chains slowed down, we were asked to find most original ways to distribute SCENESSE® under specialised transport, against the time ticking to get the hormonal therapy to hospitals on the hour. In March, northern Italy was under siege from COVID, and many porphyria patients became understandably too anxious to travel or seek treatment in designated hospitals.
It is an understatement to say 2020 has been quite a test, but not at any time have I detected a sense of panic or loss of control across our group of companies. Rather, a silent confidence underpinned our staff that they would collectively manage the uncertainty even if it meant accepting some delays. Surprisingly, during the global lockdown there was a high percentage of requests from known EPP patients for continued SCENESSE® treatment at the peak of the daily COVID hazards. As seen from our operational results, we have steadily grown the Company, despite the spread of the pandemic. In analysing the rate of continuation of SCENESSE® treatment on 30 June, we found 96.6% of patients who had started treatment in 2016 were still requesting and receiving the drug. Overall, through qualitative surveys patients have reported a positive change and or improvement in their behaviours and activities undertaken, as recorded by an inventory of daily activities. Even in 2020, the number of treatments increased year on year as well as the number of new patients.
GOVERNMENTS’ ROLE
In the wake of the global pandemic we have witnessed the rise of the state’s role in choosing which workers to defend, which companies to assist and where to allocate resources among the general population. Households in G8 countries were handed free money, while relief was given to businesses for rents and additional incentives to retain workers. In a Keynesian doctrine where the state plays a central role, central banks have kept interest rates low while state debt is now predicted to rise to 15-17% of GDP as borrowed funds among western countries. The stimulus seen in Europe, the USA and Australia seems to have no boundaries with an aim to keep economies fluid.
There are many ifs and nobody, including our central banks, really knows how to forecast our economies for the next 12 months. We are in unchartered territory.
However, I continue to believe that a deflationary environment cannot be maintained, nor can prices be kept low long-term, and when inflation eventually creeps in, the concern will be how this will affect businesses and specific sectors. Without having foresight, I see it as management’s task to read the markets as well as we possibly can and conceptualise how to protect the group of companies against these macro-economic waves.
Central banks are now openly intervening in public markets. In parallel, the state’s financial regulators find themselves overburdened in combatting distortion of equity markets where algorithms and short selling dominate daily trades. At CLINUVEL, we have had our fair share of these practices as witnessed since April 2019. Having gone through these episodes in the past (most conspicuously in 2007), I know from experience that continued corporate performance, generating further intrinsic value will eventually wash out these short sellers. In the meantime, the pain is borne by all shareholders, including CLINUVEL’s staff, ergo holding steady is the mandatory approach.
Economies are seeing the delayed effects of the pandemic, whereby the surplus nations such as Germany, Japan and China have seen a sharp decline in exports, while overall consumption has been reduced to the necessary goods and services. While the International Monetary Fund and US Federal Reserve have revised their forecast to a fall of GDP in 2021 to under 5%, the European Central Bank has been more moderate and predicts negative growth of 3% for 2021. As a footnote, the further global recovery is contingent on the fiscal and economic stimulus by the richer nations slowly waning off, central policies to remain effective, foreign demand to pick up and societal uncertainty to ebb away. There are many ifs and nobody, including our central banks, really knows how to forecast our economies for the next 12 months. We are in unchartered territory.
Reviewing 2020, chaos ruled in China first, followed by confusion on the European continent and eventually, lockdowns in the US and Australia and periods of circuit-breakers in Singapore and South-East Asia. Just when the world prepared to resume to normality in May, the second wave of the pandemic struck Europe, Australia, and parts of Asia, whereby the US had remained throughout the year in a state of nationwide restriction. At the time of going to print, there seems to be a renewed emergence of the viral spread, with an R-factor of 1.6 in the European Economic Zone and steady spread in the US. Thus, prolongation of restrictive measures seems unavoidable.
ADAPTIVE MINDSET REQUIRED
Adaptation – as in rapidly accepting a changing environment – comes to mind as the main hymn when reflecting on 2020. I have been most impressed by our teams and their interconnectedness. New professionals joining the Group were without warning asked to work from home, some of them not properly set up to work remotely and unfamiliar with the processes of the new company they had joined. Communication has become the key attribute to cross functional flow, operating procedures, and advancement of R&D, while financial discipline is maintained. Thus far, CLINUVEL’s staff have stood up to the task and the same talented pool of people will continue to rise to the challenges as the economies and markets continue to oscillate.
At the peak of the first wave of COVID, we introduced an individual monitoring system for tracking communications. The weekly reports provide us insight into the Group’s activities hour by hour, since we needed a baseline number of outward and inbound emails, video conferences and connectivity between our global teams.
With a mindset to globalisation, we concluded paradoxically that crisis management needed to take place locally. While Brexit had been postponed twice and the definitive transition period is set to end on 31 December 2020, we were forced to consolidate our UK operations while establishing two new hubs within the European Economic Area. Integration of the Group is key to continued success, meaning that the same procedures, rules, and financial rigour needs to be adopted by all subsidiaries.
Talent joined the Company, and in a remote world very little time is available to young professionals to immerse themselves in our work that is heavily dictated by deadlines and due dates. I am of the belief that young people need to be endowed with responsibilities early on and I repeat this phrase in many interviews with candidates. At CLINUVEL we have laid a basis whereby staff are accountable early on in their career, errors are accepted but a thorough evaluation and avoidance of repeat is required. In these unexpected times and economic reversal of fortunes, individual responsibility is more important than ever. I have seen rays of light and real prodigies shining through our R&D, clinical, commercial, and financial management.
The new Chairman introduced advanced measures to align all staff with the corporate’s objectives, while the Risk Charter and governance were reviewed in depth. The financial management of the Group remains one of our core strengths, the IR and PR efforts of our team reaped benefits and the media and communications team increased its activities by putting out more published news than previously through various social media channels, ASX announcements and translating technical news for broader audiences.
In the new economic era, I view radical changes to CLINUVEL’s business plan as imperative. Less will be more: less face to face interactions, fewer meetings, and faster and more effective decisions to be taken. A relaxation of risk management implies that more errors can creep in, therefore increased controls and contingency plans are needed as part of processes. For instance, in R&D one would expect less time to repeat and reproduce experiments but more time to validate methods, a greater number of formulations, more analytics run simultaneously and increased number of planning to increase the generation of replicable data. In terms of clinical trials, it will translate to more trials sponsored by CLINUVEL, more proofs of concept studies in less centres.
For the third year in a row we have issued a dividend. With the year dominated by global turmoil we wanted to demonstrate our gratitude to the long-term shareholders staying with the CLINUVEL story.
In reviewing each single crisis I have endured with our senior staff the past decades, I can confidently say that specific to CLINUVEL at each of the troubling times, new gains were found when we put our minds to original solutions. This attitude is a repetitive trait by our people, perhaps it has even become an asset of the Company, attracting to CLINUVEL new talented professionals with the same attitude. The reality of the viral threat puts the CLINUVEL team to the test – our output, financial results and handling of suppliers and hospitals during these sensitive times speaks volumes and I am proud of our staff’s resolve and perseverance exhibited during these challenging times .
I assign the current success to the discipline bonding the Company across staff and Board. Many of the features of our team over the past months have reminded me how our key managers handled the 2007-2009 crisis. It certainly gives me courage that this assembly of professionals will be able to weather unexpected events in the future. While in 2007, the loss of CUV value was due to the managers of a hedge fund dumping their portfolio of illiquid stocks on the open market, causing a steep decline in share price, the current crisis has been triggered by global events causing externalities. The management of these seismic events is not new to our team. In effect, crisis management is part of our fire-drills across many disciplines of the Group, such as in pharmacovigilance and quality control, acted out as simulation exercises.
CLINUVEL’S EXPANSION
In a discussion first initiated by analyst Sarah Mann (of Moelis Australia Securities), other colleagues had debated the value of diversification versus focus, whereby CLINUVEL has been taken as an example with a long-term business focus. I have written about this in various News Communiqués in past years. There is no absolute answer whether this is right or wrong or whether one increases inherent business risks by focussing a company on one technology and expanding onwards. Each project, each company needs to be evaluated on its own merits, as a separate business case with specific characteristics which do not lend themselves to equal comparison. However, specialisation originating from dedication and focus often leads to domination in one area of technology, science, and operations. Naturally, success attracts others to the space, and unsurprisingly CLINUVEL will have its competitors in time to come. However, focus is difficult to replicate. Diversified companies will most likely not commit to causes like focussed entities are able to do, and whether this advantage translates to long-term sustainable value will be played out in time. Furthermore, concentration on business segments generally leads to translational use of technology and more opportunities to open-up.
Under this scenario, CLINUVEL scaled up its R&D activities in-house in Singapore (VALLAURIX PTE LTD). The Singaporean government, through the Economic Development Board, recognised our investment and decided in February to contribute up to S$500,000 in funding allocated to equipment and the hiring of local personnel.
At the time of going to print, the state-of-the-art VALLAURIX Research & Development Centre has been delivered to our Group, albeit still working in shifts under the circuit breakers put in place by Lee Hsien Loong, Singapore’s ‘Chief-in-Command’. The laboratories are being equipped with modern technology, whereby both a biological and analytical laboratory are at the centre of our facilities. With this R&D hub, we have attracted a pool of talented individuals, scientists with a drive to develop Singapore’s first commercial pharmaceutical products to market. As VALLAURIX is expanding, daily coordination between our Singapore and global teams take place by various channels to ensure total integration within the Company. The VALLAURIX Research & Development Centre is part of our aim to vertically integrate all facilities in-house. This will require a further diversification of disciplines across the Group in a controlled manner. I intimated before that expansion too fast carries the risk of loss of focus and eventually failure, too slow a lack of targeted progress. Two more disciplines will be added to CLINUVEL’s armamentarium before the foundations of the house can be considered complete.
The pipeline of molecules and products coming out of VALLAURIX aims to address various markets ranging from acute diseases to over-the-counter markets complementing our current treatment and providing name recognition among wider consumer communities. The first follow-on product, the liquid formulation of afamelanotide, PRÉNUMBRA® was announced in July 2020. By working on several fronts, I am certain our research teams will continue to surprise us all with further output the next year.
Diversification from within is the mantra, and expansion of pharmaceutical products originating from the knowledge and progressive insights from our professionals is the safest way to bring CLINUVEL forward. Under the authority of Chief Scientific Officer Dr Dennis Wright, VP Scientific Affairs Dr Tim Zhao, and Head of our R&D Centre Dr Uma Rai, we have ample to look forward to. Our teams have chosen the least degree of risk in diversifying yet staying within the field of melanocortins.
In planning all the Group’s activities, Board and management is cautiously balancing receipts, profits versus investments. The past year we discreetly invested up to 20% of annual turnover in related technology and more in personnel but stayed well within our preferred financial parameters. Here, risk versus rate of progress is reviewed periodically to balance the company between prudent expansion and financial strength.
When entering a recession, the learnings from past downturns are multi-fold. Firstly, adverse market reactions last longer than most analysts predict, the economic reverberations run deeper. The current European plunge into recession will affect many sectors permanently: hospitality, travel, and catering, but also healthcare. A second take-away is that each company and business unit must keep its output high. Economic output is partially determined by the supply side, and of course the demand originating from the market served. Third, the ability to manage cash receipts obviates the need for capital, an invaluable protection of value. Although much jubilation exists about the records booked on US exchanges and the ASX, we remain cautious as to the possibility of major market corrections and its impact on shareholder sentiment. Our IR manager and finance team have had a plentiful year engaging actively with major institutions, and subsequently we have seen the emergence of two new and large US institutions on CLINUVEL’s register, holding collectively over 6%. As CLINUVEL consolidates its financial position and expands its offerings, it will attract new international investors.
For the third year in a row we have issued a dividend. With the year dominated by global turmoil we wanted to demonstrate our gratitude to the long-term shareholders staying with the CLINUVEL story. The success of this program started with a detailed planned financial strategy and under the auspices of Darren Keamy, it has been well executed. In my last period as Managing Director, I intend to see the financial management continue for the Company to secure its longevity. With that objective, we will exert our maximum effort to diversify and generate profits within the group of companies.
As Managing Director, I see it as my principal task to enable staff to excel in their performance by creating and maintaining a positive and conducive professional environment. Remaining close to my beliefs, we demand much from our staff, but take a constructive and supportive approach when it comes to the time needed to adapt and learn new skills. With that hybrid approach to the business, we pride ourselves to attract unique talent, retain staff for the longer term and thereby increase the knowhow within the Company. In 2020, there has been a balanced mix of new professionals and seasoned managers who had reached their career objectives at CLINUVEL. Some of these managers left CLINUVEL not only as accomplished professionals, but also as shareholders with substantial experience to be used in life sciences elsewhere.
At this point in time, the Company is in the best financial position since its inception.
CONCLUSION
I see it as my duty to direct an orchestra of gifted individuals, reacting and putting measures in place to secure a future for patients, staff members and shareholders in times of desperate economic conditions. The past months, we have effectively secured the supply of product and cashflow to the Company.
At this point in time, the Company is in the best financial position since its inception. The financial strategy has been executed with accuracy and expansion has supported the growth in earnings.
An increasing percentage of patients is receiving SCENESSE® treatment in more countries, and in April 2020 the distribution of the pharmaceutical product started in the United States. Our teams have been active in Europe, the United States and China, and are awaiting the Australian regulatory outcome. R&D output is forthcoming, while the second product PRÉNUMBRA® was announced in July. While I daily observe sufficient areas for improvement within the Group, we have also arrived at a point where many of us at the start of the journey could not have predicted the current position. Of course, success is a fickle definition and can be measured by various parameters such as stock price, products in pipeline and rate of expansion: higher, bigger, and more. Realistically, the success of CLINUVEL is one to build and reflect on.
I am more optimistic than ever on the future of the Company and that all our teams are collectively working towards the same objectives, (first published in 2019) outlined below:
To all those who wished us well in the weeks following the announcement of the quarterly and annual financial results, I reciprocate and wish you a healthy and prosperous year ahead in holding CLINUVEL in your portfolio. To the patients and families who have long waited for SCENESSE® in the United States, we express our appreciation for your patience. On behalf of CLINUVEL’s Board of Directors, we express our appreciation for your continued support.
MANAGING DIRECTOR’S MESSAGE TO GERMAN SPEAKING SHAREHOLDERS
Mein besonderer Dank gilt allen deutschsprachigen Aktionaeren fuer die Unterstuetzung von CLINUVEL. Wir haben von Ihnen ein gutes Feedback zu unseren deutschen Nachrichtenmitteilungen erhalten und werden diese auch weiterhin veroeffentlichen. Der diesjaehrige Geschaeftsbericht befasst sich mit Wachstum und Expansion und spiegelt den Wendepunkt in der Unternehmensgeschichte wider, der durch die Zulassung von SCENESSE® durch die US amerikanische FDA fuer die Behandlung von erwachsenen EPP-Patienten im vergangenen Oktober gekennzeichnet ist. Diese Meilensteinentscheidung ist der Katalysator, der CLINUVEL das Potenzial eroeffnet, SCENESSE® und seine Derivate auf ein breiteres Spektrum von Patientengruppen zu diversifizieren. Als langjaehrige Aktionaere wissen Sie, dass der Weg der Produktentwicklung im Pharmasektor lang sein kann. Wie in meinem Schreiben dargelegt, ist das CLINUVEL-Team entschlossen und fokussiert, diese Behandlungen fuer Patienten mit Prioritaet zum Nutzen aller voranzutreiben. Vielen Dank.
Philippe Wolgen
Managing Director, CLINUVEL Group
CLINUVEL IN THE NEW WORLD ORDER
CLINUVEL IN THE NEW WORLD ORDER
INTRODUCTION
The coronavirus pandemic has changed the world in which we live and operate. How we engage with one another, manage work and life, produce goods and services, and manage the impact of the increase in public spending incurred to support people and economic activity has changed to varying degrees and will continue to do so. The purpose of this piece is not to analyse the changes in societal norms and structural changes in economies, as this requires more time to interpret emerging trends. Rather it serves to highlight that CLINUVEL is entering a new world order in which the potential implications for people and economies are forever changed by the materialisation of a key risk – global pandemics and the way we will communicate and do business.
There is a multitude of known and unknown risks facing people, governments, businesses, and other operators in the economy. For businesses across the globe, as for other economic entities, the need for prudent risk management has been underscored by the coronavirus pandemic and its direct and indirect impact on their viability and future performance. This timely feature provides an overview of CLINUVEL’s approach to risk management and its commitment to the prudent management of Environmental, Social and Governance (ESG) criteria which are now integrated within risk management and investment decisions, and closely linked to sustainable corporate performance.
CLINUVEL’S PRUDENT APPROACH TO RISK MANAGEMENT
The prudent management of risk is essential to the achievement of CLINUVEL’s strategic initiatives and the progress of the Company towards a diverse and integrated biopharmaceutical aimed at providing treatments to multiple patient groups. CLINUVEL has a conservative risk culture and this is reflected in its thrifty financial management over the years to develop and commercialise SCENESSE® (afamelanotide 16mg) for the treatment of adult patients with erythropoietic protoporphyria (EPP). The Board of Directors has mandated its executive management to execute a conservative strategy while growing the Group of companies. Specifically, the development cost of SCENESSE® has been well below the cost of comparable pharmaceutical company orphan drug development focused on a new molecular entity. In addition, CLINUVEL’s decision to self-distribute SCENESSE® in Europe and the USA has proven cost effective and ensures value for shareholders. With careful cost control and prudent management decisions, CLINUVEL has strengthened its balance sheet after four years of commercial operations, with no debt and a cash position sufficient to finance its immediate future expansion. CLINUVEL is risk adverse and deliberate in its actions, and actively seeks to identify and proactively manage various risks.
The core of our risk approach is the maintenance of a risk register that details and ranks identified risks and their mitigation. The risk register enables the formal consideration of risk by senior management and the Board, while also highlighting potential opportunities. This in turn helps to ensure that all significant risks are suitably identified, assessed, and managed. A key part of risk management is ensuring we meet societal expectations, regulations and laws on corporate standards and conduct. Thus, since ESG criteria cover essential aspects of our operations and adherence to these serves to minimise risk, these are very much a part of our risk management approach and are included in the risk register. There is a multitude of benefits to maintaining an active risk register, the most obvious being ensuring the Company is well-run and keeping Board and management alert to the risks which need attention.
THE UNITED NATIONS GLOBAL COMPACT AND ESG
The United Nations Global Compact (Global Compact) was established in 2000 by then United Nations (UN) Secretary-General Kofi Annan to implement universal principles in business that advance responsible corporate citizenship, better aligned to the UN’s global development objectives. In January 2004, Annan wrote to 55 of the world’s leading financial institutions to invite them to develop guidelines and recommendations to better integrate ESG criteria in the operation of financial markets. Twenty financial institutions participated and endorsed the resulting report, “Who Cares Wins”, in December 2004. The report was completed under the auspices of the Global Compact and contained a “call to action” to stakeholders in the financial and business world: companies were asked to lead the implementation of ESG principles and policies by providing information and reporting on related performance and to identify and communicate the key challenges and value drivers associated with ESG issues.
Since then, ESG has gained prominence throughout the global investment community, playing a key role in the analytical assessment of companies and investment decisions. (ESG-based investment is often referred to as sustainable investing, responsible investing, impact investing or socially responsible investing.) Large and influential institutional investors have aligned prudent management of ESG criteria with sustainable long-term growth and have integrated ESG criteria into their due diligence of investments. An online search for the term “ESG and company performance” reveals a multitude of studies that support a positive correlation between responsible ESG management and enhanced financial performance, efficiency, and firm value. A review by Friede, Busch and Bassen (2015) of around 2,200 individual studies since the 1970s concluded that roughly 90% of studies found a non-negative correlation between ESG criteria and corporate financial performance, with a stable link evident over time.
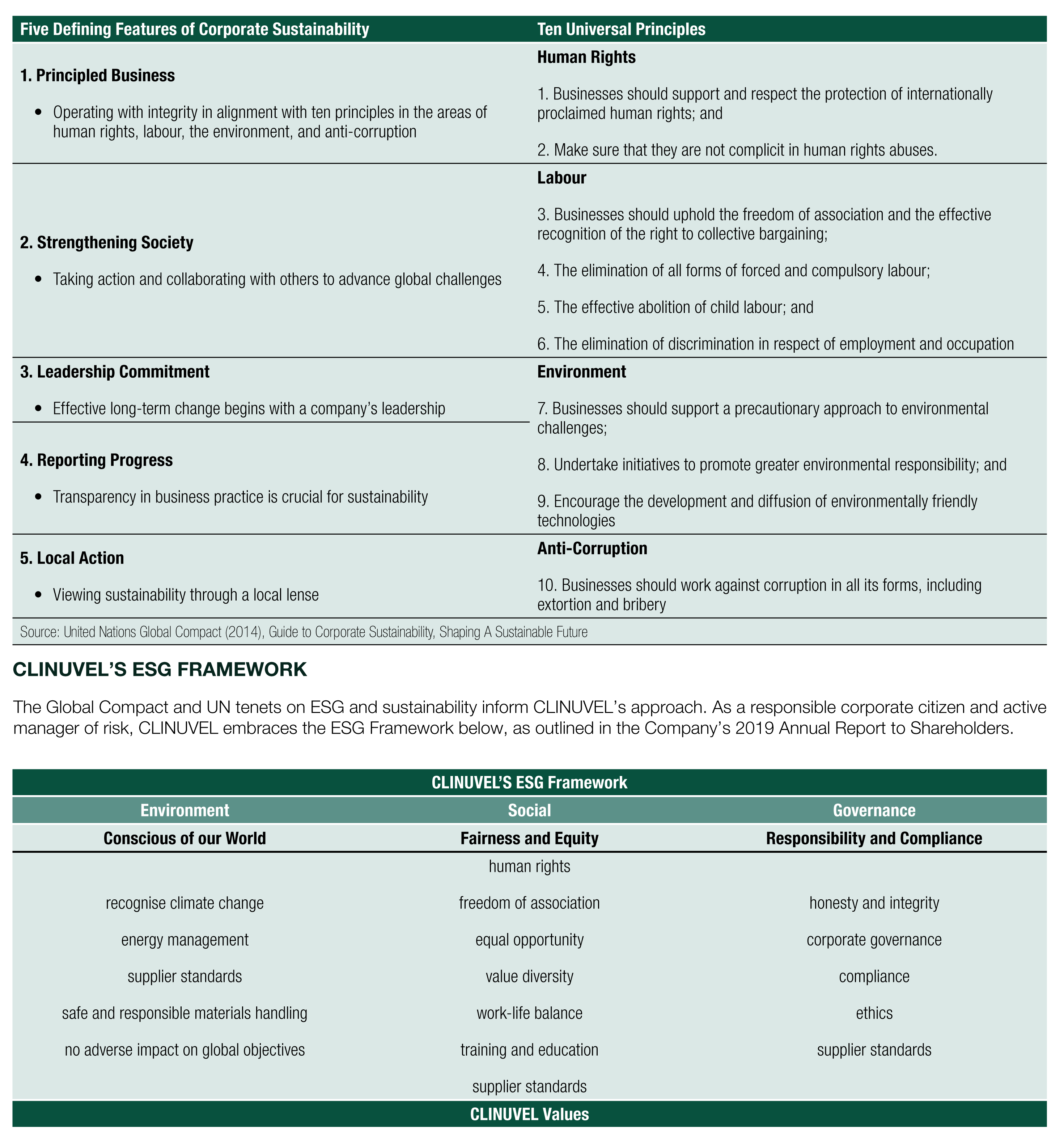
The Global Compact is a call to action to companies around the world to align their strategies and operations with five defining characteristics of corporate sustainability and ten universal principles in the areas of human rights, labour, environment, and anti-corruption. The UN vision is that appropriate action in these areas will support their broader sustainable development goals. The Global Compact is the world’s largest global voluntary corporate sustainability initiative with over 8,000 companies and 4,000 non-business participants in over 160 countries. The five defining features of corporate sustainability and ten universal principles promoted by the Global Compact are outlined below:
CLINUVEL’S ESG FRAMEWORK
The Global Compact and UN tenets on ESG and sustainability inform CLINUVEL’s approach. As a responsible corporate citizen and active manager of risk, CLINUVEL embraces the ESG Framework below, as outlined in the Company’s 2019 Annual Report to Shareholders.
CLINUVEL’s values are placed at the base of the framework to reflect their fundamental foundation in governing our behaviours and how we conduct our business. The key tenets of this framework are explained further below.
FOCUS ON THE ENVIRONMENT
We are conscious of the world in which we operate and importantly, have no adverse impact on the achievement of the UN’s global environmental objectives. Our management of environmental risk is appropriate for a relatively small company (with less than 100 employees), striving to minimise energy and water use and exercise prudence in the management of office waste as we work towards a paperless office. SCENESSE® and its key input, afamelanotide, are manufactured by reputable contactors in developed countries under World Health Organization Good Laboratory Practice (GLP) and manufacturing standards. Our new laboratory facility in Singapore is working towards ISO 17025 and GLP certification. CLINUVEL’s product SCENESSE® is administered by subcutaneous injection and dissolves in the human body and has no adverse environmental impact. We also regularly assess the commitment of our suppliers to minimise their use of scarce resources and act in and environmentally responsible manner in accordance with ESG criteria.
SOCIAL RESPONSIBILITY
CLINUVEL is a responsible corporate citizen and reflects international community standards in our policies. The UN tenets on human rights and labour standards embedded in the UN International Covenant on Economic Social and Cultural Rights (1966) and International Covenant on Civil and Political Rights (1966) are reflected in our Employee Handbook. These covenants recognise the right to work, including the opportunity to earn a living by work that is freely chosen and accepted (Article 6); the right to enjoy just and favourable conditions of work, including minimum remuneration, safe and healthy conditions of work, equal opportunities for all, rest, leisure, initiation of working hours and holidays with pay (Article 7); and the freedom of association and collective bargaining and the right to strike (Article 8). Specifically, CLINUVEL is committed:
- on Human Rights, to
- freedom of association; and
- supporting a diverse and inclusive workplace, with equality of opportunity of all employees regardless of race, colour, gender, religion, ethnicity, culture, political opinion, age, and disability.
- on Labour Standards, to
- the right to organise and collective bargaining;
- zero tolerance of child labour, forced labour and discrimination, harassment, and abuse of any kind;
- providing a decent workplace focused on the health and safety of employees;
- supporting the training and education of all employees.
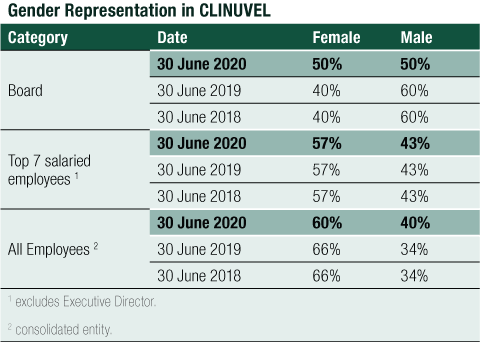
Opportunity is equal to all people in CLINUVEL and procedures are in place to escalate, review and address any human rights and labour standard concerns. The Group’s employees span 16 nationalities and gender representation is balanced, as reflected in the following table on the composition of genders on the Board, in senior management and across all employees.
Work-life balance is supported by our Employee Handbook and a new Remote Working Policy. Necessitated by the coronavirus pandemic, CLINUVEL initiated the Remote Working Policy to ensure the ongoing health and safety of all CLINUVEL employees and provides guidelines to enable an appropriate work-life balance with overall flexibility in the conduct of employees’ work, during and beyond the coronavirus pandemic. Employment security is conducted in accordance with labour laws and regulations in each of the countries in which we operate. All staff are encouraged to own an active training and development plan to support their professional fulfilment and job satisfaction.
We take a responsible approach to product development and distribution. CLINUVEL has no adverse impact on UN social objectives and makes a positive contribution through its pharmaceutical product development and distribution for unmet medical needs.
CLCLINUVEL’s research and development program is highly ethical. We undertake the minimum studies necessary to obtain the regulatory approvals required to distribute our treatments in man. We use Ethics Committees for study approval, adhere to OECD Testing Guidelines and the principles of GLP.
We are committed to the OECD Replacement Reduction and Refinement Principles for non-human studies and ensure all studies undertaken are responsibly designed and conducted by laboratories certified by internationally recognised and respected bodies. CLINUVEL disseminates its research and encourages the independent publication of the results of its clinical studies in peer-reviewed scientific journals.
A rigorous pharmacovigilance program is maintained and reported to the European Medicines Agency (EMA) in the European Union and the United States Food and Drug Administration (FDA) on the real-world experience of SCENESSE® for adult patients with EPP. SCENESSE® is carefully managed to ensure no off-label usage. Marketing and communications are aligned to the approval terms and conditions of distribution of the EMA and FDA.
CLINUVEL is responsible for ensuring that its suppliers are fit for purpose throughout the product lifecycle (from product development to commercial distribution), through risk-based quality control procedures and open communication. CLINUVEL qualifies suppliers and reviews existing relationships according to a range of expectations and standards before commencing or continuing supply arrangements.
CLINUVEL has over 15 key suppliers, most of which are domiciled in well-regulated countries that are signatories to the UN standards and objectives on sustainability and ESG criteria. CLINUVEL has a high proportion of medium- to long-term supplier relationships, enabling us to develop a good understanding of their values and commitment to ESG criteria. This diligent approach also applies to the assessment of our relationship with collaborative partners.
Should an area of concern be identified during a review of a supplier, a risk assessment is conducted to ensure the supplier is functioning within contracted capacity and standards. CLINUVEL then applies a collaborative approach with the supplier to engage in discussions with the aim of improving their qualification status and strengthening the relationship. These procedures enable CLINUVEL to maintain the qualified status of its medium to long term tenured suppliers. Furthermore, it ensures CLINUVEL’s ESG principles and those of its suppliers are consistently aligned.
CORPORATE CONDUCT AND ETHICS
Corporate governance is a key to effective risk management as it provides the policy, procedures, and compliance framework for a company’s operations. Our corporate conduct and ethics are guided by our key corporate values and governed by our Corporate Governance Protocol and annual Corporate Governance Statement.
CLINUVEL’s Board of Directors comprises the Managing Director, Dr Philippe Wolgen, and five independent Directors. They are, individually and collectively, well credentialed, with a breadth of qualifications and depth of experience in a range of fields to provide appropriate guidance to the Company. In the last year, the number of Directors has increased by one to six. The Board operates in accordance with a Board Charter that is set out in the Corporate Governance Protocol. The election and tenure of Directors is managed in accordance with the Australian Securities Exchange (ASX) Listing Guidelines. The gender representation at Board level is uniquely balanced, reflecting the broader Company. The compensation of Directors and key executives, covering key performance indicators for the assessment of short-term and long-term performance awards, are detailed annually in the Remuneration Report in the Annual Report.
CLINUVEL is focused on the maintenance of a long-term shareholder base which includes employees and a balanced composition of private, corporate, and institutional investors. CLINUVEL seeks an active and constructive dialogue with its shareholders, in accordance with ASX Listing Guidelines. We communicate frequently with shareholders, as outlined below, by operating an active program of regulatory and discretionary disclosure of information on the Company through announcements to the ASX. This includes regular corporate updates to keep shareholders informed on the Company’s performance and progress on key strategic initiatives.
CLINUVEL’s Corporate Governance Protocol meets the requirements of ASX principles and recommendations and includes a Code of Conduct and Ethics and policies on Conflicts of Interest, Share Trading and Shareholder Communications. CLINUVEL has zero tolerance of corruption of any kind. We operate a training schedule to ensure compliance with laws and regulations in the countries in which we operate. CLINUVEL staff are also encouraged to report any breaches of company values, regulations and laws and are protected from harassment and abuse on any reports of non-compliance or breaches. CLINUVEL engages with the leading law firms across the continents to ensure adherence to the highest level of governance.
CLINUVEL’s Corporate Governance Protocol sets out the code of conduct and ethics and other policies to ensure conflicts of interest are avoided and honesty and integrity prevails. We are law abiding and comply with anti-bribery and anti-corruption laws in the countries in which we operate.
MANAGEMENT OF STAKEHOLDER COMMUNICATIONS
The twenty-first century has heralded new and improved innovation in communication channels that enable the instantaneous worldwide dissemination of information – in itself, this constitutes a new world order. These advances are advantageous for the operation of capital markets and investment activities. A key part of CLINUVEL’s approach to communicating to stakeholders is the use of multiple online channels to provide up to date information on the Company’s strategy and performance. The process of dissemination starts with an announcement to the ASX, after which CLINUVEL distributes to multiple news media outlets, our ‘email updates’ list, and posts on various social media platforms. We also maintain a library of announcements and information on the Company on our website, www.clinuvel.com. However, the technological advances in communications and information exchange are not without significant potential risk to companies and orderly investment activities in general.
Information dissemination online has become the social norm and has outpaced the protection of privacy and copyright regulation and enforcement. Laws and regulatory instruments have been unable to keep up with these real-world practices and trading activities on equity markets. Identified and anonymous parties can copy and post information in breach of copyright and can make false and misleading statements, either intentionally or unintentionally, in many online channels near-simultaneously.
Parties involved in stock shorting activity actively seek to drive a company’s share price down to make a profit from share trading – they buy shares at a lower price than the shares they have borrowed to sell. Katz and Hancock (2017) of Ropes and Gray LLP note that ‘shorters’ actively use online communications either directly or indirectly through antagonists, to post reports on business platforms and discussion forums to undermine the confidence of shareholders and induce them to sell their shares at lower prices. CLINUVEL has not been immune to this type of conduct with shorting activity of CUV increasing from less than 1% of issued capital in April 2019 to a peak of 9.65% of issued capital in April 2020, before lowering to around 8.2% in late September 2020, at the time of writing this article. There has been an increase in false and misleading comment on CLINUVEL in discussion groups, which Katz and Hancock (2017) normally associate with an active shorting campaign.
CLINUVEL’s approach in the face of this activity is to focus on the progression of publicly stated strategic initiatives and provide regular and objective information to markets on the Company’s progress. This approach enables market participants to assess an objective flow of information to appropriately inform their investment decision in CLINUVEL. In the medium- to long-term, the performance of the Company should improve the demand for CUV and curtail the activity of ‘shorters’. This approach in action is reflected in the pace of the progress of the Company which has enabled more frequent material announcements to the ASX. The number of CLINUVEL announcements to the ASX increased from 44 in the year ending June 2018 to 62 and 67 in the 2019 and 2020 financial years, respectively. The Company’s discretionary announcements providing updates of the business in Chair Letters and News Communiqués are more frequent than other companies or peers in the ASX / S&P 200 Index and are authored by the Chairman and Managing Director to communicate directly to shareholders. This is a peer group leading practice. We also play a role to point regulators in the countries in which our shares are actively traded – CUV on the ASX in Australia, UR9 on the Xetra-DAX in Germany and CLVLY in the US through over-the-counter traded American Depositary Receipts administered by the Bank of New York Mellon – to inappropriate activities.
We feel a responsibility to protect key stakeholders, patients, and doctors as well as shareholders, from inappropriate online activities. This extends to us liaising with online service providers and moderators of online channels when laws and regulations are breached. There are recent indications that regulation may be starting to make up some ground in the area of online defamation with precedent setting court decisions in Australia and the United Kingdom providing clarity of what constitutes a ‘publisher’ under defamation law. Online service providers and owners of online discussion forums have been held liable as a ‘publisher’ under defamation law with the trigger of culpability being their inaction upon being made aware of defamatory posts. Interestingly, each view, like or retweet of a defamatory post is considered a new publication and individuals who anonymously post defamatory material online are at risk of being identified by online service providers and owners of online channels in response to court orders issued during pre-trial discovery. US courts have made mixed judgements depending on the circumstances of the case, but some have held online service providers liable for defamation. In the US, plaintiffs have one year to take legal action against the initial defamation and cannot claim against successive and multiple re-publications. We will not cite the sources of legal precedent here, but they can be found online. The point of these comments is to highlight that your Company is focused on reducing the risk of misinformation and breaches of the law for the sake of objective information and the protection of key stakeholders.
SUMMARY AND CONCLUSIONS
CLINUVEL has a conservative and prudent risk management culture with a firm commitment to environmental and social responsibility with appropriate corporate governance as a key accountability to underpin long-term sustainability and performance. The commitment to prudently manage risk and adhere to ESG criteria as a responsible corporate citizen is strongly held at Board level, by executive management and extends to all CLINUVEL staff. The Company’s vision and values, presented in subsequent pages of this report, support effective operations aligned to the achievement of ESG and sustainability criteria. We regard our journey to sustainability as an ongoing process of continuous review and improvement to enhance our ESG performance and to share our progress with stakeholders in periodic announcements and reports, such as this Annual Report. All stakeholders should be comforted by this responsible and prudent approach, particularly as we enter a new world order.
Sources
The Global Compact (2004), Who Cares Wins, Connecting Financial Markets to a Changing World
UNEP Finance Initiative, A legal framework for the integration of environmental, social and governance issues into institutional investment, October 2005
New York Stock Exchange (2006), Principles for Responsible Investment
Gunnar Friede, Timo Busch and Alexander Bassen (2015) ESG and financial performance: aggregated evidence from more than 2000 empirical studies, Journal of Sustainable Finance & Investment, 5:4, 210-233, DOI: 10.1080/20430795.2015.1118917
United Nations Global Compact (2014), Guide to Corporate Sustainability, Shaping A Sustainable Future
United Nations Global Compact (2017), United Nations Global Compact Progress Report, Business Solutions to Sustainable Development
International Labour Organization (2015), Compendium of International Labour Conventions and Recommendations, http://www.ilo.org/wcmsp5/groups/public/—ed_norm/—normes/documents/publication/wcms_413175.pdf
Jeff Katz and Annie Hancock, Ropes and Gray LLP (2017), Short Activism: The Risk in Anonymous Online Short Attacks, Harvard Law School Forum on Corporate Governance, November 27
INNOVATION IN DNA REPAIR
INNOVATION IN DNA REPAIR
CLINUVEL’S FOCUS ON DNA REGENERATION
The backbone of human life is created by two structures forming our genetic material: pyrimidines and purines, which in turn consist of four nucleotides adenine (A), thymine (T), guanine (G), and cytosine (C). These form the DNA-helix which carries our present and future make-up, our genetic codes. Human physiology tries to keep DNA strands intact at all costs. As we are exposed to daily oxidative stress, skin cells work hard to preserve our genetic program.
At CLINUVEL, we have focussed for two decades on the interaction of light and human biology, whereby our scientific teams have worked on replicating the deleterious effects of solar radiation through standardised conditions in our laboratories. We have used broadband ultraviolet B (UVB), narrowband UVB, ultraviolet A 1 and 2 and High Energy Visible (HEV) light to irradiate human skin and observe the instantaneous reactions and damage to the epidermis and dermis. Both in healthy and diseased individuals, skin reactions can be predicted and mapped out. In most of CLINUVEL’s clinical trials, skin surfaces of various patient groups were exposed to light sources to provoke typical sun-related symptoms. In testing the lead drug afamelanotide, we collected this data and assessed its potency for systemic photoprotection.
Going a step further, CLINUVEL evaluated its drug afamelanotide in various photodermatoses and lightinduced disorders such as polymorphic light eruption (PLE), solar urticaria (SU), acne, and Hailey-Hailey Disease, and in genodermatoses erythropoietic protoporphyria (EPP), congenital erythropoietic porphyria (CEP) and xeroderma pigmentosum (XP), all diseases where radiation at certain wavelengths play a part in the triggering of severe symptoms.
In optics and physics precision is key, and in some disorders, one specifies action spectra and the inhibition spectra at particular wavelengths causing the start of symptoms in patients. Mapping out each monochromatic wavelength along the electromagnetic spectrum is part of the disciplines of photobiology, photodermatology and radiation biology, deriving scientific knowledge from optics and physics. This knowledge has become part of our in-house strength, expertise and starting point to transgress into the area of UV-provoked DNA damage.
As genetics and cellular biology took a flight the past decennia, the knowledge of the effects of ultraviolet radiation (UVR) on human cells (skin, eyes, and organs) has increased. Most are aware of the beneficial effects of 15-30 minutes of UVR per day to stimulate the synthesis of vitamin D in our skin. However, we are also made aware of how longer periods of exposure to UVR and HEV light may lead to sunburns, photodamage, actinic damage (elastosis) and an increased risk of skin cancers as skin damage becomes chronic and permanent.
The progression from healthy human volunteers to genetic disorders has been logical for CLINUVEL, whereby the most severe diseases have deserved treatment first, not least as these stand the greatest chance of regulatory approval in global pharmaceutical markets, such as Europe, the United States, Australia and Asia. That is not to say that other disorders would not benefit from afamelanotide or melanocortins, but the development route is best justified when regulatory support is given early on in a program.
HISTORY OF DNA RESEARCH
Damage to DNA provoked by UV radiation received first attention in 1893, when Robert Bowles published an article in the British Journal of Dermatology suggesting that sunlight may be responsible for skin cancers: “If the sun’s rays will produce sunburn, erythema, eczema solare, inflammation, and blistering, it is clearly capable of producing deep and intractable ulcerations of a low and chronic nature.” This finding was corroborated one year later by Paul Gerson Unna, who associated the severe degenerative changes on exposed areas of sailors’ skin with the development of skin cancer and rapid aging. In the last three decades, various renowned research institutions have focussed on the acute and chronic damage of UV and HEV radiation to human skin. Latitudinal predispositions, localisation, skin type, family history of skin damage and skin cancers were among the variables studied. As cellular biology attracted attention the cellular signalling, expressions of genes and proteins, became a focus. As the human genome project gathered momentum at the start of the millennium, the importance of MAP kinases, endothelin, WNT, cKIT, MITF and MC1R pathways in providing skin cells with the right input, directions and stimulus became widely known.
DNA REPAIR
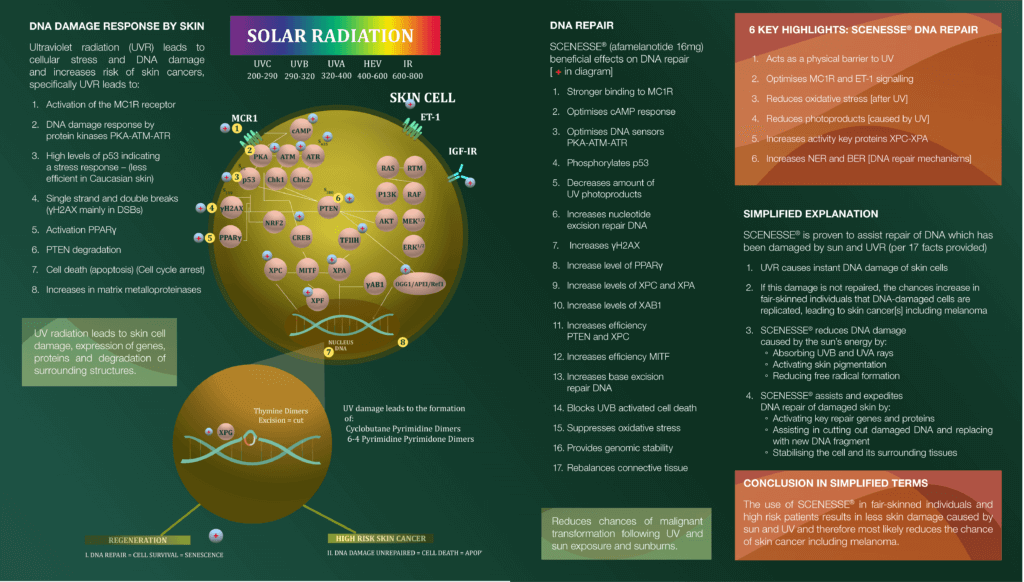
The double-page figure to this article explains the DNA Damage Response (DDR). Starting on the left-hand side, one sees the DDR by skin cells which leads to a human defence reaction. The activation of MC1R leads to a cascade of reactions seen within the cell. Human biology is fascinating in that many reactions take place simultaneously at the speed of nanoseconds. The signalling of a skin cell – there are many different types – occurs from top to bottom, starting with a particular receptor.
The PKA-ATM-ATR axis, or the communication between these intracellular proteins, causes multiple reactions further down the cell. Following solar (UV-HEV) exposure, human reactions are needed to protect cellular structures: organelles and proteins, but specifically the nucleus which may also require reinforcement. In many ways, all the process and activity taking place above the nucleus aim to protect the genomic information, DNA, within the core of each skin cell.
UV radiation can lead to double or single strand DNA breaks, although the scientific experts tend to focus on single strand breaks. UV causes typical mutations to DNA, known as UVsignature mutations, connotated as C→T or CC→TT substitutions of nucleotides within a DNA strand. Thus, we can detect and recognise the kind of DNA damage caused by solar exposure. UV also causes some of the protein expressions to be downgraded, suppressed, and dysfunctional, making the cell work at a suboptimal level, in turn causing an inferior output by the skin cell. Together, the cellular dysfunction and substitutions of nucleotides are a cause of great concern since human biology is forced to restore the balance immediately by repairing the cellular structures. Sun exposure makes skin cells work overtime. In the doublepage figure, one sees the result of this UV cascade, the formation of Cyclobutane Pyrimidine Dimers (CPDs) and 6-4 Pyrimidine Pyrimidone Dimers (6-4PPs), or photoproducts within the nucleus caused by the sun/UVR/HEV light.
Our task after each sun exposure and sunburn is to eliminate these photoproducts by replacing a piece of DNA. Under normal circumstances, the human body is quite effective at doing so, although fair-skinned individuals (skin type I, II and III) are less efficient than darker skin types (IV, V, VI). In the first group the risk of permanent mutations is increased and in these individuals the incidence of skin cancers is much higher.
While we are interested in one pathway, the breakthrough in knowledge gained the last 15 years is that various pathways and genes within a cell communicate with each other; “horizontal discussions” occur between pathways. Therefore, the dysfunction along one pathway is sought to be compensated along another, all with the aim to restore function and UV damage.
On the right-hand side of the double-page figure one reads the properties of alpha-melanocyte stimulating hormone and its reinforced analogue – afamelanotide – in its role restoring UV damage. There are more than 30 cellular domains where the protein and DNA function are aided by alpha-melanocyte stimulating hormone, and here the 17 most important ones are listed. From CLINUVEL’s data and specific scientific work performed, one expects that afamelanotide will be a determinant in the DNA reparative processes within the cell, specifically needed for those individuals at higher risk of developing skin cancers due to genetic receptors, and their ability to respond to the UV signal.
AN EXCITING FUTURE IN PROSPECT
The coming year, CLINUVEL will be furthering this field of research in human subjects (XP patients and healthy volunteers) to confirm the efficiency of afamelanotide in these cellular and DNA reparative processes.
Fitzpatrick Types l-Vl
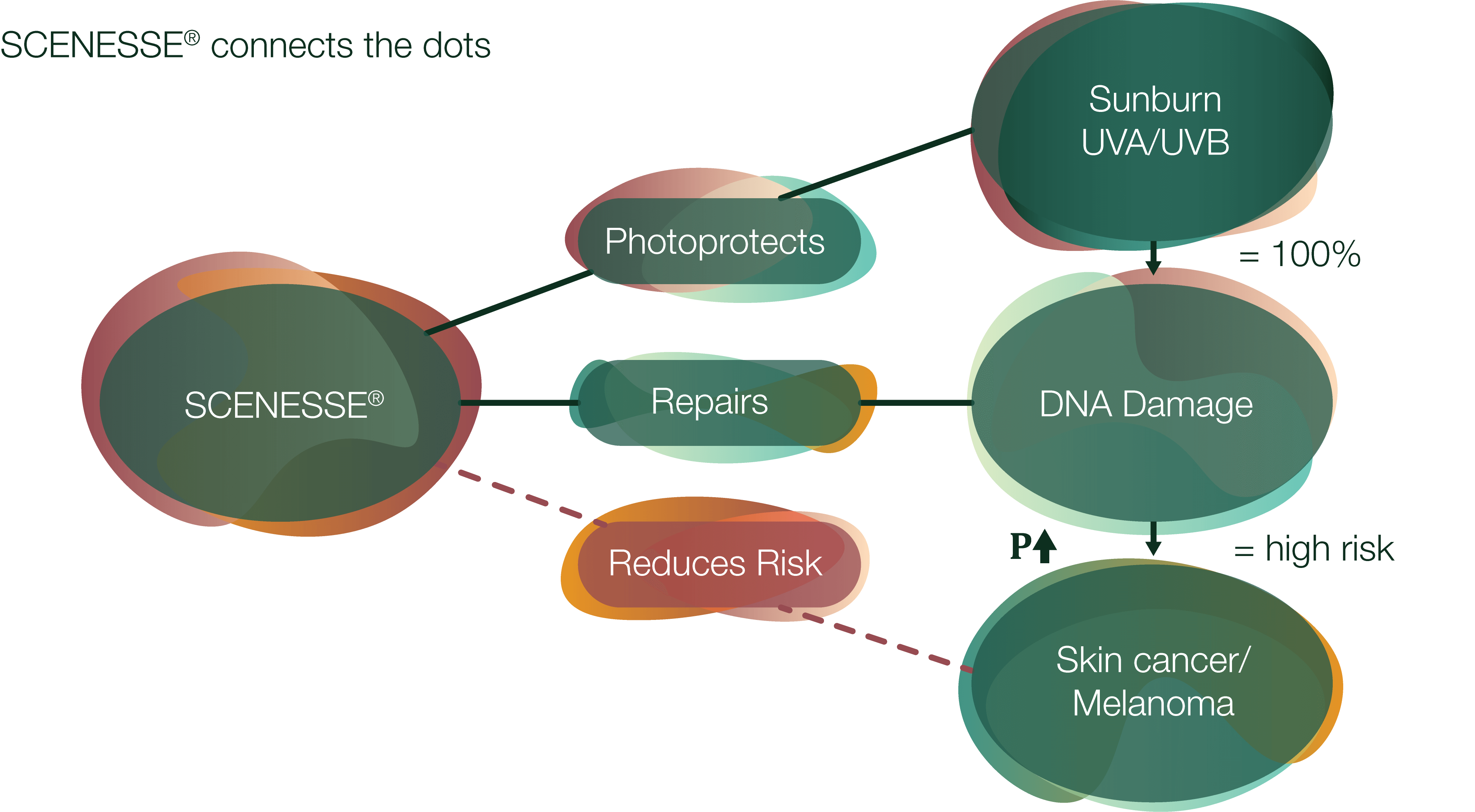
Having taken more than a decade of clinical and scientific research, it is exciting to ‘close the loop’ in using afamelanotide from:
(i) assessing UV-induced skin damage in healthy volunteers, to
(ii) systemic photoprotection in diseases patients (photodermatoses), and finally, to
(iii) reducing the risk of cellular and DNA damage caused by sun/UVR/HEV light in higher risk populations (both healthy and diseased individuals), and therefore reducing the risk of skin cancer(s).
Skin cancer comes in many forms, but the three most frequently seen in the clinic are basal cell carcinoma, squamous cell carcinoma and melanoma (various types). Common to all three forms is that UV and sun exposure play a part in the development, whereby other genetic and epigenetic factors play a role. However, the ability to eliminate or reduce the UV-inducing factor in the genesis of skin cancer is a big step forward. Hormonal therapy with a melanocortin may well be the future answer, since the use of the hormone simulates the biological function of the peptide in our body, protecting us against the insult of solar radiation. Within seconds of sun exposure, alpha-melanocyte stimulating hormone is detected as being released by our skin cells as a protective measure.
A reinforced version of alpha-melanocyte stimulating hormone was developed and formulated as afamelanotide, and the vision is to use the drug as a DNA-protectant in many formulations. The requisite is and remains the safety of afamelanotide in patients and healthy volunteers, but each day that goes by is one extra day towards the 100,000 plus patient exposure days providing evidence of safe use.
Biomimicry is the phenomenon found in biology capturing and replicating nature’s ability to provide function and protection. In afamelanotide, CLINUVEL has developed a long used potent hormone to prevent sun and light damage in those who need this most, namely, those who are at risk and those who suffer from genetic disorders affecting their ability to go outdoors. Over the next 12 months, the CLINUVEL story will unfold and the significance of our progress in DNA repair in XP and healthy individuals to the general population at risk of UV-HEV damage will become apparent.