Clinical Programs Gather Pace
CLINUVEL has built unique expertise in the development of a novel class of peptides – melanocortins and their analogues – as therapies for patient groups with high unmet medical need.
Pre-Conditions for Clinical Expansion
Several key criteria needed to be satisfied before CLINUVEL aggressively expanded its clinical program to treat new patient groups with afamelanotide and other melanocortins. The most critical criteria were the:
- US FDA approval of SCENESSE® (afamelanotide 16mg) for the treatment of adult EPP patients in the US, which ‘unlocked the door’ to CLINUVEL’s growth and expansion;
- long-term safety record of SCENESSE®; and
- accumulated expertise in the family of melanocortins and their role in key functions of the body.
The opening of the Group’s state-of-the-art Research, Development & Innovation Centre in Singapore in August 2020 was also an important pre-requisite to support more intensive molecular profiling, peptide chemistry, and polymer and formulation sciences.
It is notable, particularly in the current environment, that the Group is self-financing its growth and expansion with the cash reserves accumulated and the ongoing net cash being generated from commercial operations. The Company has not needed to raise funds from capital markets and/or financiers to enable the expansion of the clinical program.
Clinical Expansion – Step by Step
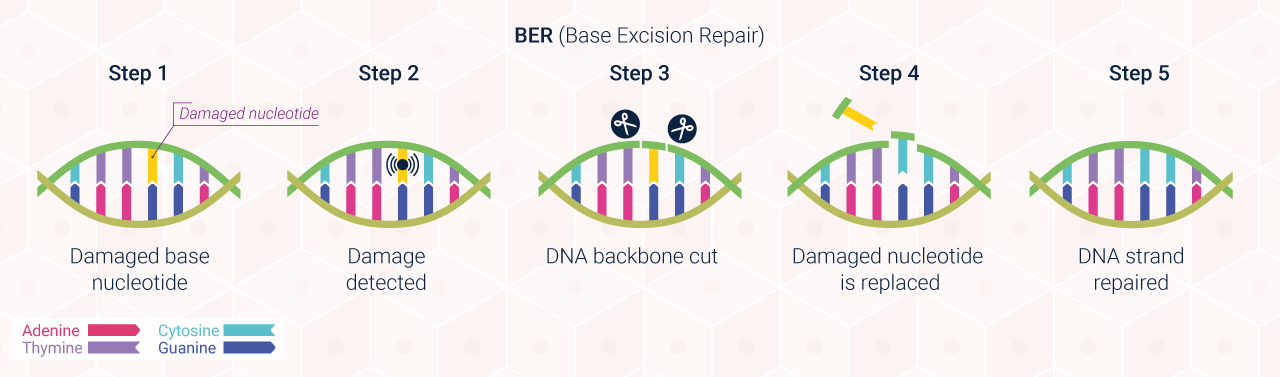
CLINUVEL is progressing clinical programs in the following key indications: DNA Repair, with an initial focus on xeroderma pigmentosum (XP); vitiligo; and arterial ischaemic stroke (AIS).
DNA Repair Program
The expanded clinical program commenced with the announcement of the DNA Repair Program in September 2020. This program is of significance to two billion people worldwide who have an impairment in their natural DNA repair processes. The initial focus of CLINUVEL’s DNA Repair Program is on XP. People with this rare and hereditary disorder are unable to repair the cellular damage to their skin caused by exposure to UV and sunlight, which is known as photodamage. This results in the mutation of their skin which leads to skin cancer at an early age. Their risk of skin cancer is over 1,000 times that of the general population, with patient life expectancy tragically around 30 years.
CLINUVEL’s DNA Repair Program initially encompasses five clinical studies involving XP patients with the XP-C and XP-V complementation, who are at the highest risk of exposure to UV and high energy visible (HEV) light, as well as disease-free individuals who serve as a control group. Three clinical studies commenced in FY2022, focusing on the safety of afamelanotide as a DNA regenerative therapy:
- CUV156 in adult XP-C patients;
- CUV151, a mechanistic study in disease free adults; and
- CUV152 in adult XP-V and XP-C patients.
These studies involve taking samples (biopsies) of exposed skin areas for laboratory analyses of DNA damage before and after drug administration. CLINUVEL has collaborated with expert physicians to develop global assessment tools and patient reported outcomes for use in the studies. In addition to confirmation of the safety of SCENESSE® in these cohorts, the objective of the studies is to see a reduction in oxidative damage in the skin biopsies after drug administration.
Read outs of these studies are expected to issue later in 2022, subject to full recruitment and completion of treatment. Pending the results of these initial studies, two further studies are planned, CUV153 and CUV154. The adjacent table summarises the studies.
When completed, the five studies are expected to collectively enrol 38 XP patients and 10 disease-free control subjects. The results will be analysed and if favourable, discussions will take place with regulatory authorities on the filing of a dossier to seek marketing authorisation on for the product for the treatment of XP. The Company will announce key milestones as the studies progress.
A proven therapy, which safely offers protection and assists DNA repair, would reduce the overall burden of the disease for XP patients and their families and could dramatically improve quality of life and life expectancy. Beyond this critical milestone for XP patients, CLINUVEL will assess other clinical indications with a view to extending the benefit of melanocortin therapy to other groups with deficient DNA repair processes. The learnings from the DNA Repair Program are also expected to be translated to the dermatocosmetic products under development for broader populations and specifically, three targeted audiences of highest risk. To learn more about this initiative, read the next feature of the Annual Report.
| Study | Patients | n = | Commenced | Objectives | First results |
|---|---|---|---|---|---|
| CUV156 | XP-C | 6 | October 2021 | Safety and reduction in oxidative damage | 2022 |
| CUV151 | Disease-free subjects | 10 | December 2021 | Reduction in oxidative damage | 2022 |
| CUV152 | XP-V and XP-C | 6 | February 2022 | Safety and reduction in oxidative damage | 2022 |
| CUV153 | XP-V and XP-C | 6 | Planned | Safety, assist DNA repair, quality of life | Not yet disclosed |
| CUV154 | XP-V and XP-C | 20 | Planned | Safety, assist DNA repair, quality of life | Not yet disclosed |

Vitiligo
Vitiligo is a common skin disorder in which the pigment producing cells of the skin (melanocytes) are absent or demonstrate a lack of activity. As a result, lighter depigmented patches of skin (vitiliginous lesions) appear on different sites across the body due the lack of melanin (pigment). The precise cause of vitiligo remains unknown. Vitiligo also causes significant psychological and emotional distress. A high disease burden is experienced by patients with Fitzpatrick Skin Types IV-VI (darker skin), where the contrasting loss of skin colour can lead to a profound sense of loss of identity.
On 8 March 2021, the FDA hosted a patient-focused vitiligo meeting for the first time, when testimonies were given of the impact of vitiligo in patients of darker skin colour, and its effect on their quality of life. The FDA acknowledged that “(t)his forum is important to the FDA, drug companies, researchers and other medical product developers that were in attendance on the day. The Agency hopes that the information provided to the FDA will help advance the science and development of new treatments for patients who would like to have them.”
CLINUVEL has demonstrated its commitment to develop a therapy focussed on the systemic repigmentation of the skin of vitiligo patients. Earlier studies, CUV102 (in the US in 2011) and CUV103 (in Singapore in 2014), have shown that treatment with afamelanotide in combination with narrowband ultraviolet B (NB-UVB) phototherapy can achieve faster and deeper repigmentation than NB-UVB as monotherapy, currently the most common treatment in vitiligo. The studies individually showed that the combination treatment led to follicular repigmentation and meaningful clinical results within six months. In the larger CUV102 study of 41 vitiligo patients, the repigmentation achieved was significant (p=0.025); refer to the picture below.
In December 2021, agreement was reached agreement with the FDA on a new Phase II study, CUV104, to evaluate SCENESSE® as a monotherapy for adult vitiligo patients. Approval to proceed with the study was received from the Institutional Review Board (IRB; ethics committee) in May 2022. CUV104 is now scheduled to commence at an expert centre in North America with the recruitment of up to six patients in the second half of 2022. The study will assess the efficacy of afamelanotide to repigmentation the face and body and improve the quality of life of adult vitiligo patients. More specifically, the endpoints of the study will be the extent and speed of repigment seen in patients as measured by the Vitiligo Area Scoring Index (VASI) tool, as well as the impact of the treatment with validated disease-specific quality of life tools. The focus of the study is on patients with darker skin types (Fitzpatrick IV-VI), more than half a million of whom are estimated to live in Europe and North America. CLINUVEL has learnt how the impact of the disease is greatest for these patients and understands from our earlier studies that they may see the greatest therapeutic response to the monotherapy treatment. Following the completion of CUV104, we will assess the results and implement the next phase of studies.
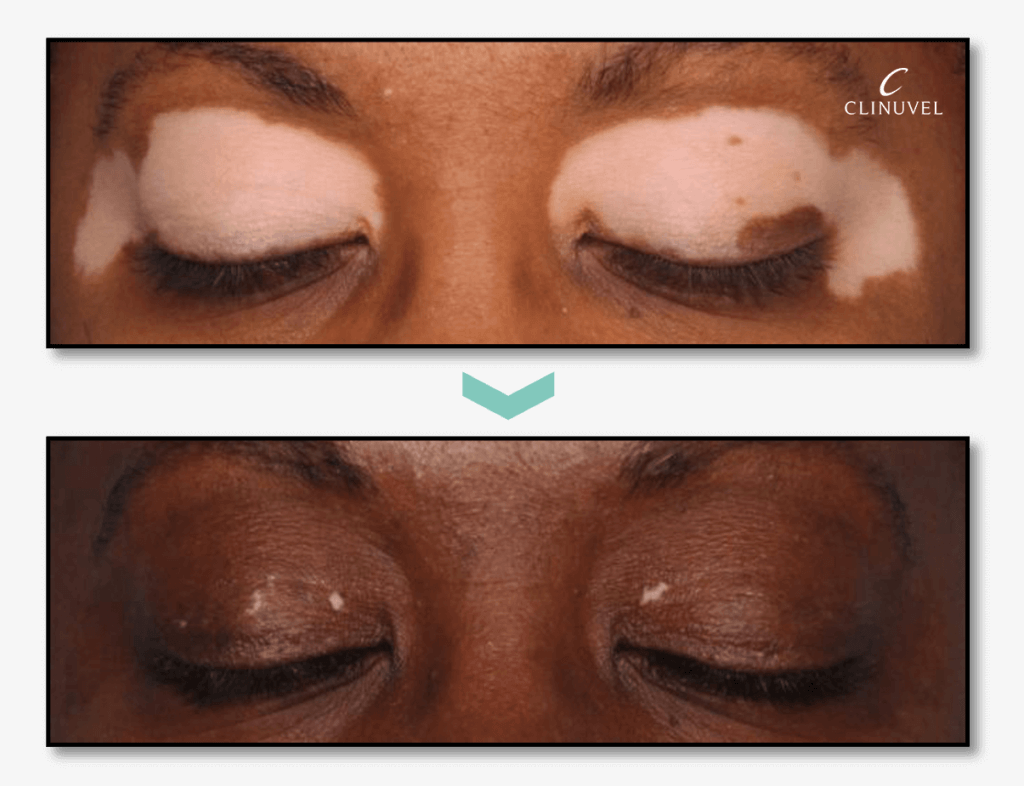
p=0.025 (VASI)
Arterial Ischaemic Stroke (AIS)
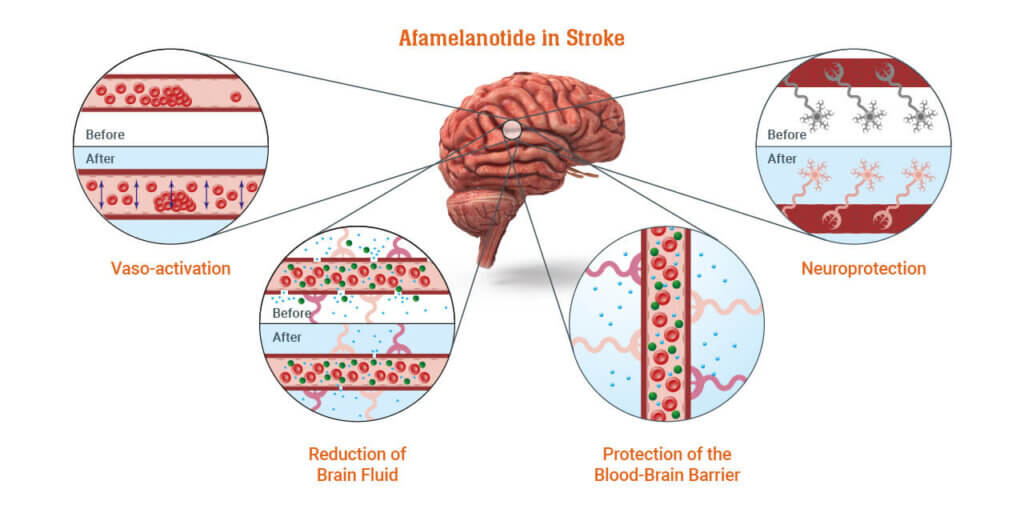
In June 2021 the Company commenced the first study of afamelanotide in AIS patients, evaluating the safety and efficacy of interventional treatment in six adult patients. Afamelanotide was administered up to four times over ten days following the stroke. The final results of the study, announced in May 2022, showed afamelanotide was well tolerated with five of the six patients experiencing considerable clinical and functional recovery up to 42 days after treatment. Specifically, National Institutes of Health Stroke Scale (NIHSS) scores improved in five patients and brain scans (MRI-FLAIR) in all patients showed a reduction in affected tissue.
Following the positive results of CUV801, CLINUVEL is commencing a new study in AIS patients, CUV803. The Company has identified stroke as the first target indication for its PRÉNUMBRA® Instant formulation, which will be used in the CUV803 study. Patient enrolment is expected to commence in the second half of 2022 pending regulatory and ethics approvals. CUV803 will maintain a focus on safety with neurological function and extent of damage, while evaluating a flexible and higher dosing regimen to CUV801. PRÉNUMBRA® Instant provides a flexible, fast-acting dosage form of afamelanotide as a subcutaneous liquid injectable.
Addressable Market of Indications
The addressable market of each of the indications being assessed is summarised below:
| Indication | Addressable Market |
|---|---|
|
DNA Repair XP* |
|
| Vitiligo |
|
| AIS |
|
* Two billion people have inefficient DNA repair processes, putting them at risk of skin cancer(s) and melanoma. There are an estimated 19.3 million skin cancer cases globally. |
|
The size of the addressable markets of the indications for which CLINUVEL is progressing treatments varies from ultra-rare to large. XP is ultra-rare, but the relevance of testing afamelanotide as an assisting agent for cellular DNA-damage repair is high, since many ‘high risk’ populations eventually develop skin cancers. Vitiligo is a sizeable problem to patients, specifically for those with a darker skin type. Collectively, the addressable markets of the clinical programs underway are large, and warrant our attempt to develop commercial solutions.
Expansion of the Drug Portfolio
The starting point of CLINUVEL’s pharmaceutical portfolio is SCENESSE®, a controlled-release formulation of the melanocortin analogue afamelanotide. It is administered as a subcutaneous, solid injectable which dissolves under the skin. With its expertise in melanocortin drug development, CLINUVEL is following a program to commercialise other formulations and products to add to dosing and treatment flexibility of an expanded range of indications with unmet needs.
PRÉNUMBRA®
In July 2020, we announced the development of PRÉNUMBRA®, liquid formulations of afamelanotide to provide dosing flexibility as part of the life-cycle management of afamelanotide and to address clinical needs in critical disorders. This is the second formulation of the active pharmaceutical ingredient afamelanotide developed by CLINUVEL. PRÉNUMBRA® is being advanced as a potent haemodynamic, vasoactive (acting on blood vessels) and anti-oncotic (counteracting fluid formation in tissues) therapeutic agent, initially in adult patients.
Product development has progressed and working under an exclusive agreement, the PRÉNUMBRA® Instant presentation is being manufactured according to current Good Manufacturing Practice (cGMP) guidelines for use in clinical studies. In July 2022 we announced that PRÉNUMBRA® Instant will be administered in the CUV803 study as a treatment for stroke (AIS), with the study expected to commence in the second half of 2022. CLINUVEL is also developing the PRÉNUMBRA® Modified-release formulation. Other potential target indications are under evaluation for PRÉNUMBRA®, including disorders of the central nervous system, and are to be announced.
NEURACTHEL®
In November 2021, we announced the addition of the adrenocorticotropic hormone (ACTH) drug substance to CLINUVEL’s melanocortin drug portfolio. To be developed as NEURACTHEL® in Instant and Modified-release formulations with application to neurological, endocrinological and degenerative diseases. In March 2022, we reported that the ACTH drug substance was being manufactured for CLINUVEL under cGMP and the development and validation work for the product formulations are ongoing.
The expanded melanocortin drug portfolio is summarised below.
| SCENESSE® (afamelanotide 16mg) | Implant | Adults – EPP, XP, vitiligo, stroke | Commercial, In development |
|---|---|---|---|
| SCENESSE® Enfance | Liquid | Paediatric 12-17– EPP, XP, vitiligo | In development |
| PRÉNUMBRA® Instant | Liquid | All ages – stroke, XP, CNS disorders | Developed for clinical use in stroke |
| PRÉNUMBRA® Modified-release | Liquid | Adults – stroke, CNS disorders | In development |
| NEURACTHEL® Instant | Liquid | Adults – acute neurological, endocrinological, degenerative disorders | Update expected Q3 |
| NEURACTHEL® Modified-release | Liquid | Adults – acute neurological, endocrinological, degenerative disorders | In development |
| CUV9900 | Topical, leave on | Adults – anti-oxidative, DNA repair | In development |
| Phimelanotide | Topical, leave on | Adults – repigmentation | In development |
| Parvysmelanotide | Topical, leave on | Adults – repigmentation | In development |
The expansion of the melanocortin drug portfolio is integral to, and supports, CLINUVEL’s clinical program.
Summary
CLINUVEL’s clinical programs generate data on the safety and efficacy of our melanocortin drugs in specific medical indications. The expansion of both the melanocortin drug portfolio and the clinical program is well advanced. This is a critical part of the Company’s growth and expansion strategy towards a diversified and sustainable pharmaceutical group.